Abstract
Objective: The efficacy,
safety and advantages of endoscope-assisted microneurosurgery were evaluated in
radical operation for spina bifida.
Methods: The
neuroendoscope (2 mm-diameter rigid endoscope) was applied during microsurgery
in fifty cases of spina bifida that were operated upon at King Hussein Medical
Centre between January 2006 and January 2008. It was used prior to the incision
of the sac in cystic cases and after the dura was opened in non-cystic cases. The
cases included nearly all types of spina bifida.
Results: The
neuroendoscope showed pathology with clarification of surgico-anatomical
structures and defined its relation to the normal structures. There were no
technical difficulties or complications. It was of value in assessing the type
of lipoma, the extent of cord involvement, identified the roots at exit from
the cord invaded by lipoma and reduced both manipulation and retraction. It
gave an excellent view of the anterior part of the malformed cord with detailed
view of nerve roots and vessels before the surgical intervention. Filum
terminale and sacral roots were visualized in all cases of tethered cord making
resection of the spinal lipoma easier and safer. There was no increase in
infection rate, no effect on the primary surgical procedure, no bleeding or
nervous tissue injury
Conclusions: Application
of the neuroendoscope to microsurgery of spina bifida is very useful in showing
the pathology with more clarity and its relation to normal structures. It can enhance
surgical quality and reduce possible complications, allowing meticulous and
more complete resection while preserving neurological function. There was a
highly magnified panoramic view of the anatomy and pathology through the neuroendoscope.
Key words: Microneurosurgery,
Myeloschisis, Neuroendoscopic observation, Spina Bifida, Spinal Lipoma.
JRMS
September 2010; 17(3): 39-44
Introduction
Visualisation in spina bifida surgery is
crucial; cord, roots, and vessels are hidden behind or within the lesion.
Identification of these structures allows meticulous and safer resection of the
lesion. Spina bifida surgery aims to prevent worsening of the patient and/or
improving neurological function. Microsurgery is the standard technique in spina
bifida surgery. Operative microscope allows better visualization of pathology
and normal anatomy. However, it is limited by the fact that the field of view
achieved is restricted to the line of sight from the lens to the lesion of
interest. The surgeon cannot look around lesions to see normal structures,
identify roots, and blood vessels which are frequently obscured by pathology.(1-6)
The endoscope offers several distinct
advantages over the operating microscope and this makes it a good adjunctive
tool during microsurgery.(7-10) High magnification gives greater definition of blood vessels, roots, nervous tissue and pathology. This magnification is achieved through the lens system, proximity to the lesion, presence of clear cerebrospinal fluid (CSF) and better illumination of structures which facilitate identification of normal structures, and specially the anatomical and pathological plane of division (relation of pathology to normal structures). The endoscope also allows looking around corners and behind structures giving an extra eye for the surgeon and reducing the need for retraction and manipulation. Three dimensional aspects become clear with endoscope usage.(11-13)
Table I. Patients’ Diagnosis
|
Case No.
|
Age
|
Gender
|
Diagnosis
|
SBNS
|
|
|
|
|
|
Pre-op
|
Post-op
|
|
1
2
3
4
5
6
7
8
9
10
11
12
13
14
15
16
17
18
19
20
21
22
23
24
25
26
27
28
29
30
31
32
33
34
35
36
37
38
39
40
41
42
43
44
45
46
47
48
49
50
|
3 days
2 days
3 days
7 days
10 days
14 days
5 days
3 months
5 months
6 months
9 months
10 months
11 months
5 months
3 months
4 years
5 years
6 years
4 years
7 years
6 years
5 years
5 years
7 years
6 years
5 years
3 years
2 years
1 year
5 years
3 years
2 years
1 year
4 years
3 years
6 years
5 years
8 years
5 years
4 years
2 years
1 year
3 months
2 months
4 months
6 years
5 years
3 months
2 years
6 months
|
M
F
M
F
F
F
F
M
F
F
M
F
F
M
M
M
F
F
M
F
F
F
F
F
F
F
M
M
F
F
F
F
M
F
M
F
F
F
F
F
F
M
F
M
F
F
F
F
M
F
|
Myelomeningocele
Myelomeningocele
Myelomeningocele
Myelomeningocele
Myelomeningocele
Myelomeningocele
Myelomeningocystocele
Lipomeningocele
Lipomeningocele
Lipomyelomeningocele
Lipomyelomeningocele
Lipomyelomeningocele
Lipomyelomeningocele
Lipomyelomeningocele
Lipomyelomeningocystocele
Spinal lipoma
(Caudal)
Spinal lipoma
(Caudal)
Spinal lipoma
(Caudal)
Spinal lipoma
(Caudal)
Spinal lipoma
(Caudal)
Spinal lipoma
(Dorsal)
Spinal lipoma
(Dorsal)
Spinal lipoma
(Dorsal)
Spinal lipoma
(transitional)
Spinal lipoma
(transitional)
Spinal lipoma
(transitional)
Spinal lipoma
(transitional)
Spinal lipoma
(transitional)
Spinal lipoma
(transitional)
Spinal lipoma
(Filar)
Spinal lipoma
(Filar)
Spinal lipoma
(Filar)
Spinal lipoma
(Filar)
Spinal lipoma
(Filar)
Spinal lipoma
(Filar)
Spinal lipoma (
C+T)
Spinal lipoma (
C+T)
Spinal lipoma (
C+T)
Spinal lipoma
(D+T)
Spinal lipoma
(D+T)
CDS
CDS
CDS
CDS
CDS
CDS
CDS
CDS
CDS
CDS
|
11
6
11
11
3
5
4
8
7
11
11
10
11
5
6
15
15
15
14
14
15
13
15
15
14
14
13
15
14
15
15
15
15
15
14
12
13
11
14
13
15
15
11
11
11
15
15
11
15
11
|
11
6
11
11
3
5
4
8
7
11
11
10
11
5
6
15
15
15
14
14
15
13
15
15
14
14
13
15
14
15
15
15
15
15
14
12
13
11
14
13
15
15
11
11
11
15
15
11
15
11
|
Endoscopic and endoscope-assisted surgery
has been used in cranial surgery. It has proved of value in surgeries of
pituitary, base skull tumours, trigeminal nerve microvascular decompression,
aneurysms, and others.(14-26)
In this study we will explore the
feasibility, safety, limitations, benefits and advantages of the neuroendoscope
as an auxiliary aid in microneurosurgery for spina bifida.
Methods
Patient
Population
A prospective study included fifty cases
that underwent radical surgery for spina bifida, between January 2006 and
January 2008, at King Hussein Medical Centre. Diagnoses spanned over the whole spectrum of
spina bifida. (Table I). Patients’ ages
ranged from one day to eight years, average of 45.9 months and 35 were females
and 15 were males. Follow up ranged from three months to two years with average
of eight months.
Endoscopic Instrumentation
A rigid neuroendoscope, with high resolution
imaging and free hand manipulation, was used.
Surgical Procedure
Routine pre-operative workup and operative
preparation included plain x-rays of the spine, lumbo-sacral 3D CT scan and
MRI. Patients were operated in the prone position. Operative procedure was
performed in the usual fashion including the parts of macrosurgery and microsurgery.
The neuroendoscope was applied according to pathology; in cystic cases the endoscope
was used prior to surgical incision, a small stab wound in the upper lateral
part of the sac was used as an entry point, extra care was taken to preserve
CSF. Upper lateral approach to the sac helps in reducing CSF loss and gives
space to manoeuvre the free hand held endoscope. In cases with no cystic part,
the endoscope was introduced when the dura was opened and an entry point
achieved. Saline provided sufficient replacement to lost CSF to keep the
endoscope immersed in a fluid surrounding.
The endoscopic exploration started with
identification of anomalous anatomy; conus, roots, vessels, and filum terminale
in the malformed structure. Tracing roots backward from the exit foramina made
identification easier. It was essential to explore all sides with proper mental recall of the
anatomical and pathological findings. The endoscope was recalled whenever there
was, doubt, or need to assess completeness or safety of resection.
Results
The neuroendoscope was used with no
technical difficulties in the performance of the procedure. No extra workup or
preparation was needed. Peri operative setting of the endoscope by assistant
did reduce the extra time needed for using the endoscope to only the actual
time of its usage in the procedure which was in average five minutes. This
further helped in reducing the time spent in microscopic exploration to
identify structures.
The use of thr neuroendoscope did not
affect the spina bifida repair adversely. There was no increase in infection
rate (no infection in all cases). There was no nervous tissue injury; the Spina
Bifida Neurological Scale (SBNS) values for all cases remained unchanged
post-operatively. No
bleeding during the endoscope use was registered. The retraction and
manipulation of the cord and roots was reduced by the use of the endoscope
In spinal lipomas the neuroendoscope showed
clearly the type and extension into the conus. The line of separation between
the lipoma and the nervous tissue was identified. Roots were visualised and
their relation to pathology determined. This allowed safer and more meticulous resection
of the lipomas.
In tethered cord, due to thickened filum
terminale and filar type lipomas, the sacral roots were localized and eventually
the filum was clearly isolated and resected under direct vision dorsally and
ventrally. In lesions associated with cystic portion, the endoscope showed the
pathology clearly. Status of the roots and the presence of lipoma were
determined.
Illustrative Cases
Case 1
Seven-day-old neonate with a myelomeningocele
in the lumbar region, his SBNS was 8/11 (motor (M):4/6, reflex(R):3/4, bladder-bowel
(BB):1/5 [infant]). Repair was done through combined microscopic and endoscopic
use. The endoscope used prior to incision of the sac showed the placode, nerve
roots and vessels. (Fig. 1 A-E).
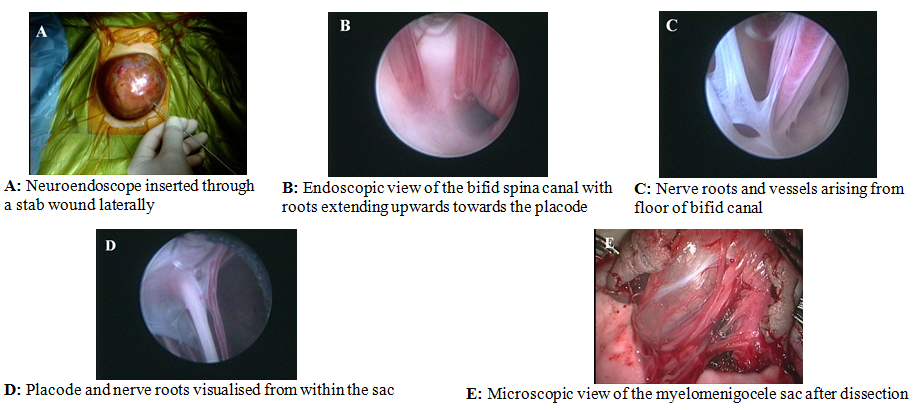
|
Fig. 1. Case 1. Images obtained during the repair in a
patient with myelomeningocele
|
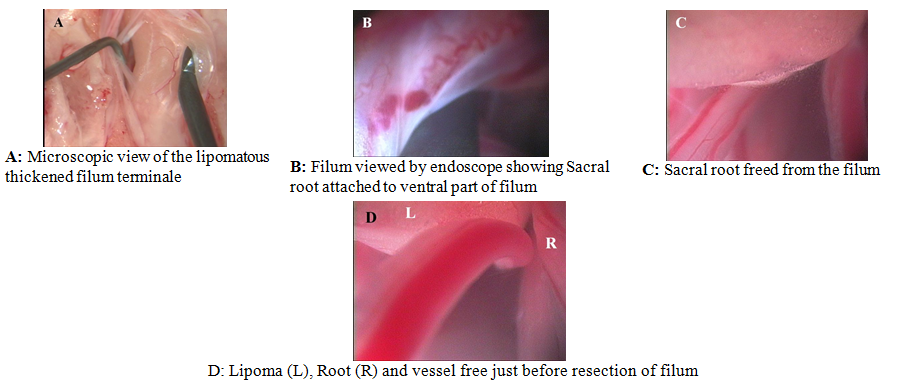
Fig. 2. Case 2. Intraoperative images obtained in a patient with
tethered cord and filar lipoma
Case 2
Seven-month-old baby with a congenital
dermal sinus. Her SBNS was 11/11 (motor (M):6/6, reflex(R):4/4, bladder-bowel (BB):1/5
[infant]). Lumbosacral MRI showed a thickened lipomatous filum terminale and the
conus at L5 level. Untethering procedure was done by endoscope-assisted microneurosurgery.
The endoscope showed the filar lipoma, sacral roots were easily visualised and
freed. Resection was done under direct vision dorsally and ventrally. (Fig. 2
A-B)
Case 3
Nine-month-old baby was referred to us with ahigh sacral dimple and lipomatous mass in the lumbar
region. Her SBNS was 11/11 (motor (M):6/6, reflex(R):4/4, bladder-bowel
(BB):1/5 [infant]). Lumbosacral MRI showed a tract extending from the dimple to the dural sac
within the lipoma which
was intra and
extradural and involving the conus, (caudal
and transitional type). Tract and
extradural part of lipoma were dissected and dura
opened,
the endoscope was used showing nerve roots on both
sides of conus, the exact
zone of separation
of lipoma and conus was
determined. Resection proceeded with very satisfactory result.
(Fig. 3A-F).
Discussion
The use of neuroendoscope in spina bifida
surgery is feasible and safe; it does not affect adversely the risk, time, or
outcome of the original surgery.
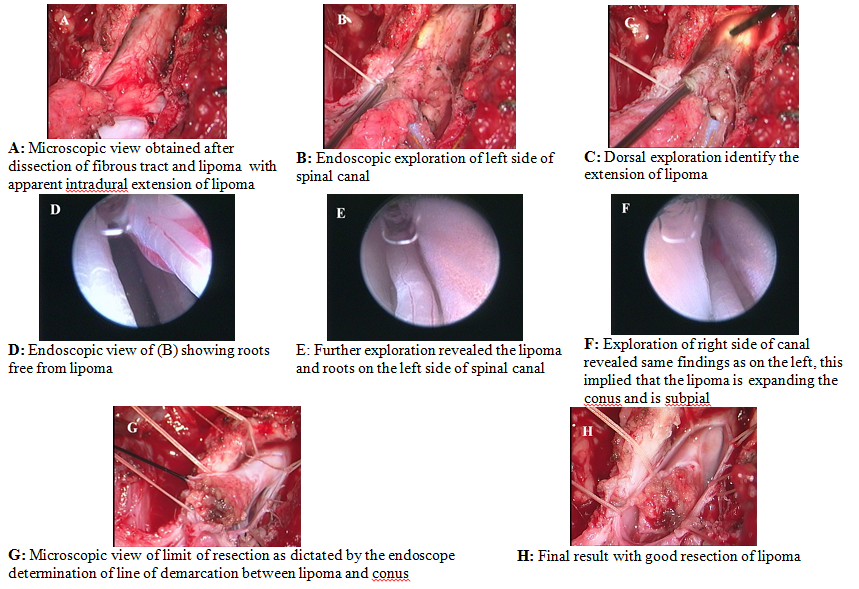
Fig. 3. Case 3. Images obtained during the radical surgery in a
patient with spinal lipoma-caudal and transitional type
Despite the belief of limitations on the
use of endoscope in the spinal canal intradurally, there were no difficulties
in using the endoscope in our cases which did cover nearly all the range of spina
bifida, spinal lipoma, and tethered cord.
The endoscope did give clearer view of the
operative field showing pathology and normal anatomical structures, especially
the anterior aspect of the cord, roots, and line of demarcation between the
lipoma and the spinal cord.
The
value of the neuroendoscope usage varies according to the type of pathology and
its complexity, the more complex the pathology the more beneficial. Spinal
lipomas were the cases of highest yield of information from the neuroendoscope.
The neuroendoscope is not to replace the microscope.
Microsurgery principles and procedures
should always be observed and practiced.
The combination of the neuroendoscope and
microscope can improve surgical results. The surgeon is no longer limited to the line
of sight between the microscope and the pathology, but can visualize details
around and behind structures. This reduces traction, manipulation, and offers
several different viewing angles. One can trace caudal and cephalic pathology
and visualise tissue under greater magnification, better illumination and
proximity.
Literature review did not show previous
similar trial to use the endoscope in spina bifida surgery, and in open dura. To our best knowledge there is no description
of this technique or its similar use.
Conclusion
Application of the neuroendoscope to
microsurgery of spina bifida is very useful in showing the pathology with more
clarity and its relation to normal structures. It can enhance surgical quality
and reduce possible complications, allowing meticulous and more complete
resection while preserving neurological function. Through the neuroendoscope
there was a new prospect of anatomy with a highly magnified panoramic view of
the anatomy and pathology.
The application of this technique coupled
with the use of intra-operative neurophysiological
monitoring could enhance the functional outcome.
References
1. Auer L
M, Holzer P. Endoscopic neurosurgery. Acta
Neurochir 1988; 90(1-2):1-14.
2. Liu CY, Wang MY, Apuzzo
ML. The physics of
image formation in the neuroendoscope. Childs Nerv Syst 2004 Nov; 20(11-12):777-82.
3. Stachura K, Libionka W. An outline of the history of
neuroendoscopy. Prezgl Lek 2007; 64(2): 118-20.
4. Kriss TC, Kriss VM. History of operating microscope: From
magnifying glass to microneurosurgery. Neurosurgery 1998 Apr; 42(4):899-907.
5. Mroczkowski E, Wielgosz R. The 80-th anniversary of the use of an
operating microscope in otorhinolaryngology. Part II. Technical principle and
operation of surgical microscope. Otolaryngol Pol 2004; 58(3):675-9.
6. Tamai S. History of microsurgery-from
the beginning until the end of the 1970s. Microneuro Surgery 1993; 14(1):6-13.
7. Fries G, Perneczky A. Endoscope-assisted brain surgery: part
2-analysis of 380 procedures. Neurosurgery 1998 Feb; 42(2): 226-31; discussion 231-2.
8.
Hopf NJ. Endoscopic neurosurgery "around the corner"
with a rigid endoscope. Technical note. Minim Invasive Neurosurg 1999
Mar; 42(1):27-31.
9.
Baron EM, Levene HB, Heller JE. Neuroendoscopy for
spinal disorders: a brief review. Neurosurg Focus 2005 Dec 15:19(6): E5.
10. Perneczky A, Fries G. Endoscope-assisted brain surgery: part
1-evolution, basic concept, and current technique. Neurosurgery 1998
Feb; 42(2):219-24; discussion 224-5.
11. Liu CY, Wang MY, Apuzzo
ML. The evolution
and future minimalism in neurological surgery. Childs Nerv Syst 2004
Nov; 20(11-12):783-9. Epub 2004 Jul 30. Review.
12. El-Shazly M,
El-Sonbaty M, Kamel A. Endoscopic-assisted microsurgery: microsurgery in the new
millennium? A comparative experimental study. Br J Plast Surg 2003 Jan; 56(1):37-40.
13. Matula C, Tschabitscher M,
Day JD.
Endoscopically assisted
microneurosurgery. Acta
Neurochir (Wein) 1995; 134(3-4):190-5.
14. Bakshi A, Bakshi A, Banerji AK. Neuroendoscope-assisted evacuation of
large intracerebral haematomas: introduction of a new, minimally invasive
technique. Preliminary report. Neurosurg Focus 2004 Jun 15; 16(6):e9.
15. Cappabianca P, Cavallo LM,
de Divitiis E.
Endoscopy and transsphenoidal surgery. Neurosurgery 2004 Oct; 55(4):933-40.
16. El-Garem HF, Bader-El-Dine
M, Talaat AM, Magnan J. Endoscopy as a tool in minimally invasive trigeminal neuralgia
surgery. Otol Neurotol 2002 Mar;23(2):132-5.
17. Gangemi M, Maiuri F,
Collela G. Endoscopic
surgery for large
posterior fossa arachnoid cysts.
Minim Invasive Neurosurg 2001 Mar; 44(1):21-4.
18. Hayashi N, Cohen AR. Endoscope-assisted far-lateral
transcondylar approach to the skull base. Minim Invasive Neurosurg 2002
Sep; 45(3):132-5.
19. Hopf NJ, Perneczky A. Endoscopic neurosurgery and
endoscope-assisted microneurosurgery for the treatment of intracranial cysts. Neurosurgery
1998 Dec; 43(6):1330-6.
20. Menovsky T, Grotenhuis JA,
de Vries J.
Endoscope-assisted supraorbital craniotomy for lesions of the interpeduncular
fossa. Neurosurgery 1999 Jan; 44(1):106-10.
21. Kasliwal MK, Mahaptra AK. Surgery for spinal cord
lipomas. Indian J Pediatr 2007 Apr; 74(4):357-62.
22. Murad GJ, Clayman M, Seagle
MB.
Endoscopic-assisted repair of craniosynostosis. Neurosurg Focus 2005 Dec;
15:19(6):E6.
23. Profeta G, De Falco R,
Ambrosio G, Profeta L.
Endoscope-assisted microneurosurgery for anterior circulation aneurysms using
the angle-type rigid endoscope over a 3-year period. Childs Nerv Syst
2004; 20(11-12):811-5.
24. Kitamura T, Taramoto A. The clinical efficacy of neuroendscopy in
surgical treatment for deafferation pain. J Nippon
Med Sch 2000 Feb; 67(1):13-7.
25. Myeong JK, Soo HY. Tethered spinal cord with double spinal lipomas. J
Korean Med Sci 2006; 21:1133-5.
26. Pitkin RM. Folate and neural tube defects. Am J Clin Nutr
2007 Jan; 85(1):285s-288s.
27. Oi S,
Matsumoto S. A proposed grading and scoring system for spina bifida: Spina
Bifida Neurological scale (SBNS). Childs Nerv Syst 1992; 8(6): 337-42.