Objective: To use three-dimensional ultrasound to quantify changes in junctional zone morphometry during the normal menstrual cycle in patients with unexplained infertility.
Methods: A longitudinal, observational study has been carried out in 23 women with unexplained infertility and 24 for fertile women as controls. Subjects were scanned by vaginal three-dimensional ultrasound every other day from day three of the cycle until ovulation. After confirmation of ovulation, 3D transvaginal ultrasound was then performed every four days until the next cycle. Virtual Organ Computer-Aided Analysis (VOCAL™) was used to define and measure thickness and volume of the subendometrial junctional zone at NURTRE clinic at Queen’s Medical Centre at the University of Nottingham, UK.
Results: Junctional zone volume and thickness demonstrated significant changes with time (P<0.01) in both groups of the study. There was a significant difference (P<0.01) in junctional zone thickness between fertile and subfertile groups during the postovulatory period (+1 to +9 day relative to ovulation). However, there was no significant difference (P>0.05) in junctional zone volume between both groups of the study throughout the menstrual cycle although the measurement tended to be higher in the fertile group at every point of the cycle.
Conclusion: This study has analysed dynamic morphometry of the uterine junctional zone during the normal menstrual cycle using three-dimensional ultrasound in patients with unexplained infertility. We found that there was significant growth restriction in junctional zone thickness during the implantation period in subfertile group that might explain one of the causes of infertility.
Key word: Junctional zone thickness and volume, Unexplained infertility, Three-dimensional ultrasound, VOCAL™
JRMS August 2007; 14(2): 38-44
IntroductionUnexplained infertility is defined as failure to conceive in a couple for which no definitive cause for infertility can be found.(1) Unexplained infertility affects 16% of couples.(2) There are many proposed theories about the aetiology of unexplained infertility including: pituitary and/or follicular dysfunction, gamete dysfunction, immunological factor and alteration in endometrial function with impaired subendometrial and endometrial development.(3)
The diagnosis of unexplained infertility is possible when all standard clinical investigations (semen analysis, assessment of ovulation and normal luteal phase, demonstration of tubal patency) yield normal results.(4) Three-dimensional ultrasound offers the gynaecologist a non-invasive tool to follow follicular growth, endometrial, subendometrial development and uterine blood flow.(5) It is possible that a cohort of women with unexplained infertility have decreased endometrial and subendometrial junctional zone perfusion that may lead to failed blastocyst implantation due to sub-development of these uterine tissues.(6,7)
The human myometrium is structurally and functionally polarized during the reproductive years.(8) The endometrium and subendometrial myometrium which are of paramesonephric duct origin are concerned mainly with sperm transportation, preparation of the uterus for blastocyst implantation, nourishment and protection of pregnancy, and menstruation,(8-10) whereas the outer myometrium is of non-mullerian, mesenchymal origin and mainly concerned with parturition.(11) Sub-endometrial myometrium corresponds to the junctional zone of the uterus and differs structurally and functionally from the outer myometrium.(9) Junctional zone has higher smooth muscle density, increased nucleo-cytoplasmic ratio of myocytes and higher vascularity than the outer myometrium.(12,13)
Vaginal video-sonography is a new imaging technique, which allows semiquantitative assessment of myometrial contractility waves.(10,14,15) The uterus contracts autonomously, rhythmically, and unceasingly throughout the menstrual cycle.(16)
Several authors have proposed a role for retrograde inner myometrial contractions in sperm transport through the uterine cavity and for the conservation and nourishment of preimplantation blastocystis within the upper part of the uterine cavity during the follicular and periovulatory phases of the menstrual cycle.(10,14) Higher junctional zone (JZ) contractility and subsequently increased mobility of the endometrium may impair its receptivity and affect early phases of implantation.(17,18)
Abnormal uterine junctional zone contraction pattern and dysperistalsis could be induced by abnormal development of this myometrium part. Furthermore, documentation of the endometrial wave pattern and subendometrial morphometry might contribute to understanding of conditions such as subfertility, dysmenorrhoea, endometriosis and recurrent abortion.(18) The existence of an abnormal wave pattern in the junctional zone in a subfertile patient could be another uterine factor explaining subfertility. Other examinations, such as endometrial biopsy, studying the texture of the endometrium by US, and hysteroscopy to reveal the presence of intrauterine lesions, contribute little to the understanding of the cause of otherwise unexplained infertility.(19)
Brosens et al, found that junctional zone hyperplasia in symptomatic subfertile women have a significant loss of both propagated antegrade contraction waves during early menstruation and retrograde waves in the late follicular phase when compared to controls with normal junctional zone appearance on magnetic resonance imaging. Furthermore, it appears that dysperistalsis or hypoperistalsis during menstruation contributes to menorrhagia; while pre-ovulatory hypoperistalsis may affect sperm transport through the uterus and tubes, and compound the fertility problems observed in these patients.(16)
Most studies on junctional zone thickness and pattern were done using magnetic resonance imaging, and most of these studies were done on abnormal uterus, not including whole the menstrual cycle, and on a small number of women. Apparently, more information in detail about the normal cycle changes of the uterus, the endometrium, and the junctional zone are necessary.
Therefore, in the present study we used three-dimensional transvaginal sonography in normal spontaneous ovulatory cycles in healthy fertile women and women with unexplained infertility, to describe in detail the morphological changes in junctional zone thickness and volume.
Methods
This study was a longitudinal, observational study with an experimental design. Subjects were scanned by three-dimensional transvaginal ultrasound every other day from day three of the cycle until ovulation. Blood samples also were taken. Ovulation was confirmed by the collapse of the dominant follicle and by observation of luteinizing hormone surge concentrations in the plasma. After confirmation of ovulation, three-dimensional transvaginal ultrasound was then performed every four days until the next menstrual cycle. The dates of each scan were then reclassified to plus and minus days from ovulation.
PopulationControl group: Twenty-four fertile women were recruited as a control group. Recruitment was facilitated through regional and local advertisement.
The selection criteria were as follows: the patient should be of the reproductive age (without specifying the upper or lower age), having a regular menstrual period without menstrual dysfunction or obvious subfertility, having no history of gynaecological disease or medication that may affect the uterine physiology, having no current pregnancy (they were asked to use barrier methods of contraception or abstain from intercourse throughout the study).
The exclusion criteria were: irregular menstrual cycle; menstrual disorder that needs any medication; current hormonal contraception, intra-uterine contraception device or tubal ligation; history of subfertility; pelvic inflammatory disease; endo-metriosis; polycystic ovarian disease or recurrent miscarriage.
Unexplained subfertile group: Twenty-three consecutive women with unexplained subfertility were recruited through the hospital infertility clinic and via advertisement in the local media. The selection criteria were: Ultrasonographic and endocrinological demonstration of ovulation, confirmation of tubal patency, and partner seminal analysis within the World Health Organization guidelines.(20)
Tubal patency was determined by either laparoscopy and hydrotubation or hysterosalpingo-contrast-sonography (HyCoSy) with the positive contrast agent Echovist™. During recruitment we did not restrict recruitment to those women with normal pelvis by laparoscopy, we excluded women with a history of pelvic or abdominal surgery, previous pelvic inflammatory disease and clinical features suggestive of endometriosis including heavy, painful periods and deep dyspareunia.
Subjects recruited into the study (control and subfertile) provided both written and verbal consent to participate in this study without any financial reward. The study was approved by the Local Ethical Committee.
Data acquisition: All data were acquired with ultrasound Voluson™ 530D machine (GE Kertz, Zipf, Austria) and a 7.5 MHz transvaginal probe. At the end of the scan session, the acquired volumes were loaded from the magnetic optical disk and sent to a personal computer via a dedicated Digital Imaging and Communication in Medicine link (DICOM). 3D View™ software was used by the personal computer to receive and store the volume datasets on CD for later review and analysis
Data analysis: All measurements were done by using the multiplanar display and were undertaken by a single observer (A.A) to limit bias. Three-dimensional endometrial and junctional zone volumetric were undertaken with the Virtual Organ Computer-aided AnaLysis imaging program (VOCAL™) version 4.0 within 3D View (Fig. 1). VOCAL allows us to define the volume of the endometrium and the junctional zone manually with a standard computer mouse as the data set is rotated about a central axis. For the purpose of this study, all measurements were conducted manually in plane C (coronal image) as this plane was rotated about the longitudinal plane.
Junctional zone volume measurement: After measurement of endometrial volume, we identified the area between the inner and outer myometrium (hypoechogenic area), which represents the subendometrial junctional zone of the uterus and was then traced in an anti-clock-wise fashion with the computer mouse until completion of 180˚ of rotation and the generation of the calculated volume of junctional zone including the endometrial volume (total junctional zone volume) (Fig. 1).
For the junctional zone volume calculation we subtracted the endometrial volume from the total junctional zone volume. For the purpose of this study measurement done one time in the 30º rotational angles that generated six planes for measurement because the degree of measurement reliability was high, a single measurement was considered adequate.
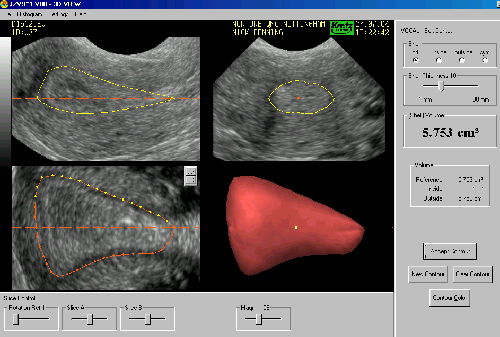
Fig. 1: The rotational technique of junctional zone volume with Virtual Organ Compute-Aided analysis (VOCAL).
The dataset has been rotated through 180° about a central axis. The border between the inner and outer myomertium manually delineated in the coronal plane. Longitudinal plane in the upper left, transverse plane in the upper right and coronal plane in the lower left of the image are shown. The resultant three-dimensional model is shown in the lower right of the image as the total junctional zone volume.
Junctional zone thickness measurementJunctional zone thickness also was measured in C-plane. Because the junctional zone is irregular in its shape and difficult to distinct the outer border, virtual line was drawn between two points, the centre of the internal cervical os and the fundal portion of the uterus. Lines were then drawn at one-third and two-third of the distance perpendicular to the initial line. Five locations on these lines were defined and measured between the hyperechogenic endometrium and mild echogenic outer myometrium (upper fundal, upper anterior, upper posterior, lower anterior, and lower posterior). The mean of these five measurements was calculated and was considered as junctional zone thickness.
Statistical analysis The appropriate statistical analysis was conducted using the Statistical Package for the Social Sciences (SPSS Release 10.1.4; SPSS Inc., Chicago, IL, USA). Data correlation was examined using Microsoft Excel and the significance of each relationship assessed by regression analysis. The type of relationship was described using SPSS.
Repeated samples analysis of variance (ANOVA) was used for the clinical studies to examine the effect of time and treatment within and between each group. Chi-squared analysis was used to test for the significance of the distribution of variables and a students’ t-test performed to examine the difference between the means when two sets of variables were present. The data presented, as the mean ± one standard error of mean (SEM). P<0.05 was considered to be statistically significant.
Results
Patients profile: The median duration of the menstrual cycle overall was 29 days for the control group and 28 days for the subfertile group with no significant difference (P>0.05). There were no significant difference between subfertile group and control group with regards to patient age and body mass index, with significant difference (P<0.05) in gravidity, parity and miscarriage.
Control Population: The final control group, which consist of 24 women, had a mean age of 31 years (range 20 to 42), a median parity of one with eight nulliparous and 16 parous women and contained twelve current smokers. All eight nulliparous women were under 31 years of age whilst of the 16 parous women, seven were under 31 and the remaining 9 were aged 31 or more. The median duration of the menstrual cycle overall was 28 days equally divided between the luteal and follicular phase.
Junctional zone morphometry during the normal menstrual cycle: Junctional zone volume increased significantly during the follicular phase (P<0.05) reaching a plateau at day one after ovulation and remaining relatively stable for further four days then decreased to reach a nadir at the end of luteal phase. Also junctional zone thickness increased significantly during the follicular phase and reaching a peak at day 1 after ovulation then decreased slowly but remained stable during the luteal phase (Fig. 2).
Effect of unexplained infertility on junctional zone morphometry: There was no significant difference (P>0.05) in junctional zone volume between the two groups of the study throughout the menstrual cycle although the measurement tended to be higher in the fertile group at every point of the cycle.
The junctional zone volume in both groups demonstrated significant changes with time (P<0.01) with junctional zone volume growth being restricted to the proliferative phase, reaching its peak on the first day after ovulation when the mean junctional zone volume was 1.5 ± 0.27 cm³ in the control group and 1.35 ± 0.32 cm³ (P>0.05) in the subfertile group. After that there was a slow decrease in junctional zone volume after day one of ovulation in fertile group, whereas there was a sharper but not significant (P>0.05) decline in subfertile group.
Both groups then showed a comparable decline in junctional zone volume to reach a nadir at the end of luteal phase reaching 0.6±0.16 cm³ (Fig. 3a).
Although there was no significant difference (P>0.05) in junctional zone thickness overall cycle (-11 to +13 day relative to ovulation) between both groups, however, there was a significant difference (P<0.01) in the junctional zone thickness between fertile and subfertile groups during postovulatory period (+1 to +9 day relative to ovulation) and there was a trend to be significantly different (P=0.055) during the periovulatory period from –1 to +1 day relative to ovulation.
The junctional zone thickness in both groups demonstrated significant changes with time (P<0.01) and the measurement tended to be higher in the fertile group at every point. Junctional zone growth being restricted to the proliferative phase and one day after ovulation where the mean junctional zone thickness was 3.10±0.37 mm in the control group and 2.88 ±0.42mm (P>0.05) in the subfertile group. Growth remained relatively stable during the luteal phase when the mean junctional zone was 2.79±0.4mm in the control group and 2.49±0.36mm (P<0.01) in the subfertile group (Fig. 3b).
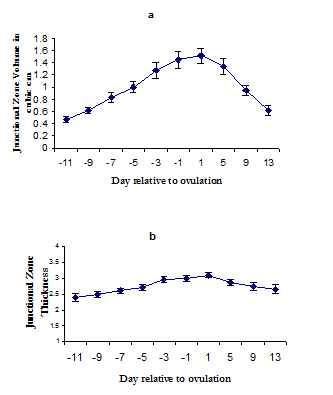 Fig. 2:
Fig. 2:Junctional zone development during the menstrual cycle in control group: Changes in junctional zone volume (a) and thickness (b) across the normal menstrual cycle in the control group of the study. The central point represents the mean value and the error bars a single standard error
Fig. 3: Effect of subfertility on junctional zone morphometry: The changes in junctional zone volume (a) and thickness (b) in fertile and subfertile groups across the menstrual cycle relative to the day of ovulation is shown. The central point represents the mean value and the error bars a single standard error.
Discussion This study, to our knowledge was the first to measure junctional zone thickness and volume from three-dimensional ultrasonography data throughout the normal menstrual cycle. We examined 470 junctional zone volume and thickness measurement in 47 women to see if there is any link of junctional zone morphometry development differences between fertile women and women complaining of unexplained infertility. All data were analysed with respect to the actual date of ovulation, determined by ultrasound and confirmed by serology of the surge in luteinizing hormone.
Our study showed that there was a significant difference (P<0.05) in the junctional zone thickness between both groups of the study during the postovulatory period until the day nine after ovulation being higher in fertile group. Growth of junctional zone thickness remained relatively stable during the luteal phase with significant difference between both groups where the mean junctional zone thickness was 2.79±0.4 mm in the fertile group and 2.49±0.32mm in the subfertile group.
Our study also showed that there was a constant increase in junctional zone volume and thickness during the proliferative phase and on the day one after ovulation in both fertile and subfertile subjects and demonstrated significant (P<0.01) changes with time in both groups. The rise in junctional zone volume reached its peak on the first day after ovulation when the mean junctional zone volume was 1.5±0.12cm³ in the fertile group and 1.35±0.10cm³ in subfertile group (P>0.05).
The bulk of information reproduces volume estimates from three-dimensional data obtained from magnetic resonance imaging; therefore there are few data with which to compare our results for junctional zone thickness and no data to compare junctional zone volume. Furthermore, none of studies of junctional zone dynamic morphometry have been performed in subfertile women during the menstrual cycle. The junctional zone thickness varies from study to another but there is a general agreement that the junctional zone thickness is variable but less than 5mm.(16,21)
Our results agree with previous published junctional zone thickness data on fertile patients. Wiczyk et al performed magnetic resonance imaging scans on five volunteers with normal ovulatory cycles on days 4, 8, 12, 16, 20, and 24 of the cycle.(22) They showed an increase in junctional zone thickness throughout the menstrual cycle in conjunction with endometrial thickness, with greatest growth from days eight and 16 as evidence for hormonal responsiveness of the junctional zone. Although our data showed marked discrepancy in mean values between the two studies with much lower mean value junctional zone thickness measurement seen in control group the pattern of growth was similar, mainly occurring between days 8 and 16 (from 2.6+0.2mm to3.1+0.3mm versus 5.1± 0.7 to 6.7 ± 0.7 mm). However, this study has its limitations that included a small number of subjects therefore no statistical analysis was carried out on the data. Furthermore, their assessments of endometrial and junctional zone were done on predetermined days rather than according to the time of definite ovulation.
Our study showed different results compared to other studies. We found that the mean value of junctional zone thickness was 2.79± 0.4 mm during late proliferative phase and 3.1± 0.3mm during the midluteal phase in fertile group. Kunz et al. found that the mean value of junctional zone thickness was 3.5 mm during the proliferative and luteal phase in healthy subjects compared to 6.5mm in women with endometriosis by using vaginal ultrasound.(15) Also Donald et al measured the junctional zone thickness using both ultrasound and magnetic resonance imaging during the early phase and late phase of normal menstrual cycle in twelve normal women and concluded that the mean thickness of the junctional zone was significantly greater with magnetic resonance imaging than with ultrasound in all pairs of examination.
There was no significant difference between the early- and late- cycle thickness of the junctional zone for either modality 4.5, 3.9mm by using magnetic resonance imaging and 2.2, 2.2mm consecutively by using vaginal ultrasound.(23) However, these studies have their limitations that included a small number of subjects and their assessment of endometrial and junctional zone were done on predetermined days rather than according to the time of definite ovulation.
Our study also showed that the junctional zone was significantly (P<0.01) thicker in fertile group during postovulatory period (+1 to +9 day relative to ovulation) with a mean value of 2.79 ± 0.4mm in fertile group. Subdevelopment of the junctional zone thickness during implantation period may have a negative impact on implantation in patients with unexplained infertility. These results agree with previous published junctional zone thickness data done by Lesny et a.l who evaluate the uterine zonal anatomy during IVF-ET using transvaginal ultrasound, they found that the junctional zone thickness was significantly (P< 0.01) thicker at the time of embryo transfer in the pregnant group, the double thickness of junctional zone was 4.2± 1.5 mm in non-pregnant versus 5.1± 1.1 in pregnant group. They concluded that the responsiveness of the junctional zone seems to be associated with implantation, and its measurement at the time of embryo transfer can be used to predict treatment outcome.(17)
Non-gravid uterus has continuous myometrial contractions that emanate only from junctional zone. Cervico-fundal contractions have a role in sperm transport and for the conservation and nourishment of the preimplantation blastocysts within the upper uterine cavity.(10) Sub-developmental growth in junctional zone may disrupt these contractions and may lead to failed blastocysts implantation, therefore can explain some causes of subfertility in patients with unexplained infertility.(16)
ConclusionThis study has analysed the dynamic morphometry of the uterine junctional zone during the normal menstrual cycle using three-dimensional ultrasound in patients with unexplained infertility. We found that there is a significant retardation in junctional zone development reflected by its thickness during the implantation period in subfertile group that might explain the cause of infertility.
Junctional zone data from three-dimensional ultrasound may play an important role in future studies investigating subfertility, menstrual disorders, and pathological changes. Three-dimensional ultrasound offers a new, reliable, non-invasive, safe and simple tool, with which to investigate uterine tissue development and could potentially be used to clarify pathophysiological changes associated with unexplained infertility and to quantify the effect of different treatment options on unexplained infertility.
Acknowledgment
The author would like to thank Dr. Raine-Fenning N. and Dr Campbell B. for their supervision and guidance during the study.
References
1.
Stewart JA. Stimulated intra-uterine insemination is not a natural choice for the treatment of unexplained subfertility: Should the guidelines be changed? Hum Reprod 2003; 18(5): 903-907.
2.
Collins JA, Crosignani PG. Unexplained infertility: a review of diagnosis, prognosis, treatment efficacy and management. Int J Gynecol Obstet 1992; 39: 267-275.
3.
Omland AK, Fedorcsak P, Storeng R. et al. Natural cycle IVF in unexplained, endometriosis-associated and tubal factor infertility. Hum Reprod 2001; 16(12): 2587-2592.
4.
Aboulghar M, Mansour R, Serour G, et al. Controlled ovarian hyperstimulation and intrauterine insemination for treatment of unexplained infertility should be limited to a maximum of three trials. Fertil Steril 2001; 75(1): 88-91.
5.
Nargund G. Time for an ultrasound revolution in reproductive medicine: Ultrasound Obstet Gynecol 2002; 20: 107-111[Editorial].
6.
Goswamy Rk, Williams G, Steptoe PC. Decreased uterine perfusion – a cause of infertility. Hum Reprod 1988; 3(8): 955-959 [Abstract].
7.
Kupesic S, Kurjak A. Uterine and ovarian perfusion during the periovulatory period assessed by transvaginal color Doppler. Fertil Steril 1993; 60(3):439-443[Abstract].
8.
Brosens J, De Souza NM, Barker FG. Uterine junctional zone: Function and disease. Lancet 1995; 346:558-560.
9.
Noe M, Kunz G, Herbetz M. The cyclic pattern of the immunocytochemical expression of oestrogen and progesterone receptors in human myometrial and endometrial layers: Characterization of the endometrial-subendometrial unit. Hum Reprod 1999; 14(1): 190-197.
10.
Lyons EA, Taylor PJ, Zheng XH, et al. Characterization of subendometrial myometrial contractions throughout the menstrual cycle in normal fertile women. Fertil Steril 1991; 55 (4): 771-774.
11.
O’Rahilly R. Prenatal human development. In: Wynn RM, ed. Biology of the Uterus, 2nd Ed. New York: Plenum Press, 1967; 35-57.
12.
Tetlow RL, Richmond I, Manton DJ, et al. Histological analysis of the uterine junctional zone as seen by transvaginal ultrasound. Ultrasound Obstet Gynecol 1999; 14: 188-193.
13.
Kelly SM, Sladkevicius P, Campbell S, Nargund G. Investigation of the infertile couple: a one-stop ultrasound-based approach. Hum Reprod 2001; 16(12): 2481-2484.
14.
De Vries K, Lyons EA, Ballard G, et al. Contractions of the inner third of the myometrium. Am J Obstet Gynecol 1990; 162(3): 679-682.
15.
Kunz G, Beil D, Huppert P, Leyendecker G. Structural abnormalities of the uterine wall in women with endometriosis and infertility visualized by vaginal sonography and magnetic resonance imaging. Human Reprod 2000; 15(1): 76-82.
16.
Brosens J, Barker FG, deSouza NM. Myometrial zonal differentiation and uterine junctional zone hyperplasia in the non-pregnant uterus. Hum Reprod Update 1998; 4(5): 496-502. Lesny P, Killick SR, Tetlow RL, et al. Uterine junctional zone contractions during assisted reproduction cycle. Hum Reprod Update 1998; 4(4): 440-445.
17.
Ijland MM, Evers J, Dunselman G, et al. Relation between endometrial wavelike activity and fecundability in spontaneous cycles. Fertil Steril 1997; 67(3): 492-496.
18.
Taylor P, Collins J. Unexplained Infertility. Oxford University Press, Oxford, United Kingdom 1992.
19.
World Health Organization. World Health Organization Laboratory Manual for the Examination of Human Semen and Sperm-Cervical Mucus Interaction. 4th ed. New York, Cambridge University Press 1999; 60-61.
20.
Mark AS, Hricak H, Heinrichs LW, et al. Adenomyosis and leiomyoma: differential diagnosis by means of magnetic resonance imaging. Radiology 1987; 163(2): 527-529[Abstract].
21.
Wiczyk HP, Janus CL, Richards CJ, et al. Comparison of Magnetic resonance imaging and ultrasound in evaluating follicular and endometrial development throughout the normal cycle. Fertil Steril 1988; 49(6): 969-972.
22.
Mitchell DG, Pannell GP, Schnholz L, et al. Zones of the uterus: discrepancy between ultrasound and magnetic resonance imaging. Radiology 1990; 174: 827-831.