Abstract
Objectives: To describe the mammographic and sonographic
features of male breast diseases, and to correlate the radiological,
cytological and histopathological diagnoses.
Methods: This is a retrospective descriptive study that was
conducted at King Hussein Medical Centre, Amman,
Jordan between January 1st 2004
and December 31st
2007. The mammograms and
breast ultrasounds of 88 symptomatic male patients were reviewed and
analyzed. A total of 24 patients with
unilateral breast masses underwent fine-needle aspiration, eight of them with
suspected malignant lesions underwent further true cut biopsy and surgery. The radiological, cytological and histopathological
diagnoses were correlated.
Results: Sixty one (70%) patients had gynaecomastia,
15 (17%) had fatty breasts (pseudo-gynaecomastia), eight (9%) had primary
breast carcinoma, two (2%) had lipomas, one (1%) had abscess, and one (1%) had hematoma. The characteristic radiological features were
confirmed by fine-needle aspiration cytology in 16 patients and by both cytology
and histopathology studies in eight cases.
Conclusion: Radiological findings
provide characteristic diagnostic appearances for certain important male breast
diseases. The radiological features can
be accurately correlated with pathological diagnosis.
Key
words: Male Breast, Mammography, Sonography
JRMS
March 2010; 17(1): 57-61
Introduction
Breast disorders in males can be distressing and patients
often feel embarrassed and anxious.(1) In our community a male patient with breast
enlargement or mass will be very reluctant to seek medical advice as this might
be considered a social stigma and a sign of incomplete masculinity. Gynaecomastia and breast cancer are the two
most common diseases of the male breast.
Other breast diseases arise from the skin and subcutaneous tissues as
fat necrosis and lipoma.(2)
Male breast cancer is rare being 1% of all breast tumors, and frequently
associated with gynaecomastia.(3) Delay in diagnosis can
result from ignorance of the existence of breast cancer in males, and this may adversely
affect prognosis. In evaluating the
clinically abnormal male breast, mammography and ultrasound are essential, and
should be performed along with the physical clinical examination.(2) The aim of this study is to describe the
radiological features of male breast diseases at King Hussein Medical Centre, and
to correlate
the radiological, cytological and histopathological diagnoses.
Methods
This is a retrospective descriptive study that was conducted at King Hussein Medical Centre, a tertiary referral hospital, Amman,Jordan from January 1st 2004 to December 31st 2007. Eighty-eight male patients referred to the Radiology department with breast complaints were reviewed in this study. Breast complaints included swelling, palpable masses, pain, and tenderness. All patients were examined by both mammography and breast ultrasound (BUS). Mammographic mediolateral oblique (MLO) and craniocaudal views were obtained for each breast using Siemens Mammomat 2 Mammography Unit. Mammograms were reviewed for the presence of gynaecomastia, masses, calcifications, lymph node enlargements, and nipple and skin changes. BUS was performed using 5-11 MHz linear transducer, ATL Philips HDI 5000 Ultrasound was done to evaluate the site, shape, outline, and echogenicity of any mass, and to observe the presence of enlarged axillary lymph nodes. Fine-needle aspiration (FNA) was collected from 24 patients presenting with unilateral breast masses, and the cytology findings were recorded. Eight patients with suspected malignant lesions underwent further true cut biopsy and surgery. The radiological appearances were described and correlated with the pathological diagnoses.
Table
I. The distribution of mammographic
patterns in Gynaecomastia
|
Gynaecomastia
|
Patients No (%)
|
Dendritic
|
Diffuse
|
Nodular
|
|
Bilateral
|
34 (56%)
|
22 (36%)
|
8 (13%)
|
4 (6%)
|
|
Unilateral
|
27 (44%)
|
15(25%)
|
5 (8%)
|
7 (11%)
|
Table
II. Symptoms of male patients presented
with Gynaecomastia
|
Clinical
Presentation
|
Total
patients No 61 (100%)
|
|
Palpable
mass
|
24
(40%)
|
|
Pain
and tenderness
|
16
(26%)
|
|
Diffuse
breast enlargement
|
13
(21%)
|
|
Swelling
and tenderness
|
8
(13%)
|
Results
A total of 88 symptomatic male patients were
investigated. The age of the patients
ranged between 25-80 years with a mean of 53 years, 61 (70%) patients had
gynaecomastia, 15 (17%) had fatty breasts (pseudo-gynaecomastia), eight (9%) had
primary breast carcinoma, two (2%) had lipomas, one (1%) had abscess, and one
(1%) had hematoma. The final diagnosis
was based primarily on typical radiological findings in 64 patients. However, radiological features were confirmed
by FNA cytological diagnosis in 24 patients: 14 with gynaecomastia, eight with
breast cancer, one with breast abscess, and one with breast hematoma.
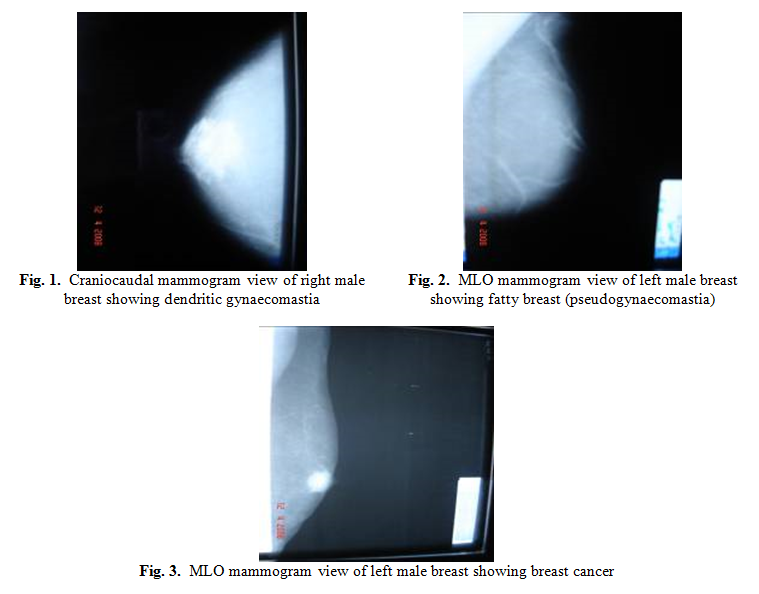
Gynaecomastia was diagnosed by mammography in 61 (70%)
patients when there was increased retroareolar density extending from the
areola in a flame-shaped (dendritic) (Fig. 1), nodular or diffuse pattern. Gynaecomastia was bilateral in 34 (56%)
patients and unilateral in 27 (44%), and the distribution of mammographic
patterns of gynaecomastia is shown in Table I.
The age of patients with gynaecomastia ranged between 25-70 years. The clinical presentations of patients with
gynaecomastia (Table II) included palpable mass in 24 (40%) patients, pain and
tenderness in 16 (26%), diffuse breast enlargement in 13 (21%), and swelling
and tenderness in eight (13%). Out of 61
patients with gynaecomastia, 14 underwent FNA, seven were on spironolactone
diuretic therapy, two on oestrogen therapy for prostate cancer, and one had
testicular carcinoma.
A total of 15 (17%) patients had fatty breasts
(pseudo-gynaecomastia) demonstrated by mammography as adipose tissue without
retroareolar density or ductal structures (Fig. 2). Those patients had no history of drug intake
or other medical illnesses; however 4 had recent increase in body weight.
Eight (9%) patients, who presented with a painless
breast mass, had radiological features consistent with primary breast
carcinoma, and the diagnoses were confirmed by FNA cytological findings. Furthermore these eight patients underwent true
cut biopsy and surgery, and the diagnoses were confirmed by histopathological
studies. They were also followed up on
annual basis. The mean age of breast
cancer patients was 68 years.
Mammography revealed central retroareolar masses with irregular ill-defined
margins (Fig. 3). There were no
microcalcifications detected in seven
patients,however
diffuse microcalcifications were shown in one patient. BUS showed an irregularly-outlined hypoechoic
retroareolar mass with posterior acoustic shadowing in five patients, and a well-circumscribed
smooth retroareolar mass in three. One mass
had prominent microcalcifications seen as bright echoes. Histological examination revealed invasive
ductal carcinoma in six patients, invasive ductal carcinoma with ductal
carcinoma in situ (DCIS) in one, and an isolated poorly differentiated DCIS in one
patient.
BUS of two patients, who presented with palpable mass,
revealed hyperechoic lesions consistent with lipomas; however the mammogram
showed a thin radio-opaque capsule of the lipoma in one patient, whereas no
abnormality was detected in the other.
The mammogram of a 70-year-old patient, who presented
with a subareolar tender swelling, revealed a lobulated mass with indistinct
borders, and BUS showed an irregular inhomogeneous mass with posterior acoustic
shadowing. These radiological features
were consistent with subareolar breast abscess and the diagnosis was confirmed
by FNA cytological findings.
The mammogram of a 45-year-old patient, who presented
with a palpable mass, revealed a well-circumscribed mass; however BUS showed
that the mass had heterogeneous echogenicity with posterior acoustic
enhancement. FNA cytological findings of
this lesion confirmed the diagnosis of hematoma.
Discussion
The normal male breast consists predominantly of fat,
and contains few secretory ducts. On
mammography, it is homogenously radiolucent with few strands of ductal or
interlobular connective tissue, without suspensory ligaments of Cooper.(4) Breast enlargement that results from increase
in fat as in obesity is called pseudo-gynaecomastia or fatty breast. This is easily differentiated from
gynaecomastia by mammography that demonstrates adipose tissue only without
retroareolar density or ductal structures.
Gynaecomastia is the benign enlargement of ductal and
stromal components of male breast tissue, resulting from a relative increase in
oestrogen effect which stimulates duct development, or decrease in androgen
effect which antagonizes the effect of oestrogen.(1)
Physiological gynaecomastia occurs in three distinct
groups: neonatal, pubertal, and adult males between the ages of 50-80. Overall, 65-90% of all male neonates have
breast tissue, resulting from the transfer of maternal and placental oestrogen
and progesterone, which persists up to several months.(1,5) At puberty, by the age of 14, up to 60% of
boys have gynaecomastia, secondary to imbalance in the normal androgen-oestrogen
ratio. This usually resolves within 1-2
years. Gynaecomastia is found in 32-65%
of healthy men at all ages. In elderly
men, gynaecomastia may be secondary to decreased testosterone production due to
testicular insufficiency.(1,6,7)
Non-physiological gynaecomastia develops with a
variety of syndromes, drugs, and diseases.
Gynaecomastia is associated with the recovery stage of starvation,
ambiguous genitalia, and Klinefelter’s syndrome. Diseases associated with gynaecomastia
include hyperthyroidism, renal disease and hemodialysis, liver cirrhosis, and primary
and secondary hypogonadism. Neoplasms producing oestrogen such as testicular
Leyding cell, sertoli cell, testicular germ cell, and adrenal tumors are also
associated with gynaecomastia. Drugs
associated with the development of gynaecomastia can be divided into
non-hormonal drugs like cimetidine, digoxin, diuretics, phenytoin and tricyclic
antidepressants, and hormonal drugs like androgens, estrogens and estrogen
agonists, choriogonadotropins, and anti androgens.(1,5,8)
In this study, seven patients with gynaecomastia were
on spironolactone diuretic therapy, two on estrogen therapy for prostate
cancer, and one had testicular carcinoma.
Clinically, gynaecomastia may be detected as a
palpable subareolar mass, usually bilateral and sometimes painful, however it
might be unilateral.(2) Three mammographic patterns of
gynaecomastia have been described. Nodular pattern appears as a fan shaped
density radiating from the nipple, and might appear spherical.(5,9) Dendritic gynaecomastia appears as a
retroareolar soft tissue density with prominent extension that radiates into
the deeper adipose tissue. Diffuse
gynaecomastia has mammographic appearances similar to a heterogeneously dense
female breast.(9,10) Cooper
and coworkers(5) reviewed mammograms of 263 male patients and
detected gynaecomastia in 213 (81%) patients, where 103 (48%) had nodular
pattern, 82 (38%) dendritic, and 28 (13%) diffuse. In another study,(2)
mammograms of 236 male patients were reviewed and detected gynaecomastia in 206
(87%) patients, where 72 (35%) had dendritic pattern, 70 (34%) nodular, and 64
(31%) diffuse. In our study, gynaecomastia
was detected in 61 (70%) patients, where 37 (61%) had dendritic pattern, 13
(21%) diffuse, and 11 (18%) nodular.
Sonography is useful in evaluating gynaecomastia, and
detecting cancers obscured by the dense breast tissue.(2) Ultrasound was recommended by Daniels and
Layer(11) as the first line imaging of gynaecomastia, and
mammography may be added to confirm the diagnosis.
Male breast cancer is less common than gynaecomastia,
and accounts for 0.5-1.0% of all breast cancers, and 0.17% of all male
carcinomas.(12) It
occurs at any age, but the mean age is 65 years. Gynaecomastia is not a risk factor for
cancer, but both can co-exist together in high estrogen status, as in men with
Klinefelter’s syndrome who have 58 folds high risk to develop breast cancer.(12-14) A family history of breast cancer increases
the risk of developing cancer in males, and this may be linked to mutation in
the BRCA2 (Breast Cancer Type 2 susceptibility protein) gene. Ashkenazi Jews have a higher prevalence of
BRCA1 (Breast Cancer Type 1 susceptibility protein) and BRCA2 genes, and an
increased risk of breast cancer than other populations.(1,12,15) In our study, the wife of one of the breast
cancer patients was his second degree relative and had breast cancer for four
years. There was an increased risk of
developing breast cancer in patients with prostate cancer receiving estrogen
therapy.(16) Exposure
to radiation, obesity, and alcohol consumption are other suggested risk factors
for male breast cancer.(8,12,15)
As with women, the most common symptom of male breast
cancer is a painless lump, and other symptoms include pain, bloody nipple
discharge, nipple retraction, and skin thickening. Breast tissue in women is largely in the
upper outer quadrant; however it is subareolar in men. That is why the tumor site for breast cancer
in men is usually subareolar.(2) Out of 87 cases reviewed by Yap et al.(16)
77 (88%) were subareolar with nipple involvement. A similar prevalence of subareolar tumor was
seen in the series of Dershaw et al.(13) The margins of the lesion may be well
defined, ill defined, or spiculated. The
lesion may be rounded, oval or irregular, and might contain numerous tiny
calcifications. In our study, all eight
breast tumors were subareolar, diffuse microcalcifications were seen only in
one patient, and there was no breast cancer detected in any patient with gynaecomastia.
BUS helps in the correct local staging of the tumor,
by identifying the degree of infiltration of the skin and pectoral muscle.(17) Yang et al. in their study on the
sonographic features of eight male breast carcinomas, reported a complex cystic
mass in four cases.(17)
Approximately
85% of breast cancers in men are infiltrating ductal carcinoma. DCIS associated
with infiltrating ductal carcinoma was found in 35-50% of male breast cancers. Pure DCIS without associated infiltrating
ductal carcinoma is a rare disease, representing approximately 5% of all male
breast cancers.(18,19)
Lobular carcinoma has been reported in only a few cases.(20) In our study, invasive ductal carcinoma was
diagnosed in six patients, infiltrating ductal carcinoma with DCIS in one, and an
isolated poorly differentiated DCIS in one. However lobular carcinoma was not detected.
On mammography, a lipoma can be shown as a circumscribed
radio-lucent lesion with a thin opaque capsule.
In our study, lipoma was detected in two patients, seen on BUS as a well
defined hyperechoic mass; however only one showed the thin radio-opaque capsule
on mammography.
Subareolar abscess is a chronic lesion associated with
duct ectasia, which tends to recur, unless treated by excision of both the
abscess and the duct.(2)
Appelbaum et al.(21) described two cases of
breast abscess, as a nodule with indistinct borders, and punctate
calcifications. There was one case of diagnosed
breast abscess in our study.
Other uncommon male breast diseases such as
tuberculosis, neurofibroblastoma, intracystic papilloma, subcutaneous leiomyoma,
and adenomyoepithelioma were not detected in our study.
Conclusion
The male breast is rudimentary, and physiologically
non functional, however it may be involved in many pathological conditions. Mammography and ultrasound are essential in
evaluating suspected male breast disease following clinical examination.
References
1.
Niewoehner
CB, Schorer AE. Gynaecomastia and breast cancer in men. BMJ
2008; 336 (7646): 709-713.
2. Günhan-Bilgen
I, Bozkaya H, Ustün EE, Memis A. Male breast disease: clinical, mammographic,
and ultrasonographic features. Eur J
Radiol 2000; 43 (3): 246-255.
3.
Sciacca
P, Benini B, Marinelli C, Borrello M, Massi G. Cancer of the
male breast. Minerva Chir 2000;
55(5): 307- 312.
4.
Michels
L, Gold RH, Arndt RD. Radiography of gynecomastia and other
disorders of the male breast. Radiology
1977; 122 (1): 117-122.
5.
Cooper
RA, Gunter BA, Ramamurthy L. Mammography in men. Radiology 1994; 191 (3): 651-656.
6.
Nuttall
FQ. Gynecomastia as a physical finding in normal
men. J Clin Endocrinol Metab
1979; 48 (2): 338-340.
7.
Gikas
P, Mokbel K. Management of gynaecomastia: an update. Int J Clin Pract 2007; 61(7):
1209-1215.
8.
Meguerditchian
AN, Falardeau M, Martin G. Male breast carcinoma. Can J Sur 2002; 45(4): 296-302.
9. Chantra
P, So G, Wollman J, Bassett L. Mammography of the male breast. AJR Am J Roentgenol 1995; 164 (4):
853-858.
10. Dershaw D. Male mammography. AJR Am J Roentgenol 1986; 146 (1):
127-131.
11. Daniels IR, Layer GT. Gynaecomastia.
Eur J Sur 2001; 167 (12): 885-892.
12. Fentiman IS, Fourquet A, Hortobagyi
GN. Male breast cancer. Lancet 2006; 367 (9510): 595-604.
13. Dershaw DD, Borgen PI, Deutch BM,
Liberma NL. Mammographic findings in men with breast
cancer. AJR Am J Roentgenol 1993;
160 (2): 267-270.
14. Swerdlow AJ, Schoemaker MJ, Higgins
CD, Wright AF, Jacobs PA, UK
Clinical Cytogenetics Group. Cancer incidence and mortality in men with
Klinefelter syndrome: a cohort study. J
Natl Cancer Inst 2005; 97(16): 1204-1210.
15. Krause W. Male breast
cancer - an andrological disease: risk factors and diagnosis. Andrologia 2004; 36 (6): 346-354.
16. Yap HY, Tashima CK, Blumenschein GR, Eckles NE. Male breast cancer: a natural history study. Cancer 1979; 44 (2): 748-754.
17. Yang WT, Whitman GJ, Yuen EH, Tse GM,
Stelling CB. Sonographic features of primary breast cancer
in men. AJR Am J Roentgenol 2001;
176 (2): 413-416.
18. Anderson WF, Devesa SS. In situ male breast carcinoma in the
Surveillance, Epidemiology, and End Results database of the National Cancer
Institute. Cancer 2005; 104(8):
1733-1741.
19. Zakhireh A, Saltzstein EC, Terreros
DA. Ductal carcinoma in situ (DCIS) of the male
breast. Breast J 2004; 10(3):
263-264.
20. Nance KV, Reddick RL. In situ and
infiltrating lobular carcinoma of the male breast. Hum Pathol 1989; 20 (12): 1220-1222.
21. Appelbaum AH, Evans GF, Levy KR,
Amirkhan RH, Schumpert TD. Mammographic appearances of male breast
disease. Radiographics 1999; 19 (3):
559-568.