Abstract
Objective: To examine the association between hyperhomocysteinemia
and the risk of coronary heart disease and to highlight the relation between
hyperhomocysteinemia and other risk factors of coronary heart disease including
smoking, hypertension, and hypercholesterolemia.
Methods: A total of 45
patients with coronary heart disease and 35 healthy controls of either sex,
aged 60 years or less, were examined. Blood samples were obtained from all
subjects at fasting and 4 hours after a methionine-loading test. The risk for
hyperhomocysteinemia and its relation to other risk factors were examined by
logistic regression analyses.
Results: Sixty percent of the
patients had hyperhomocysteinemia (fasting and postload) as compared to 40% of
the controls. The odds ratio for coronary heart disease in patients with
elevated fasting and postload homocysteine was 1.85 (C.I = 1.3-2.5, p=0.00) and
1.24 (C.I =1.1-1.39, p=0.00) respectively. No interaction between
hyperhomocysteinemia and other conventional risk factors was observed. The
likelihood of a coronary heart disease event increased approximately by 2-fold
in patients with elevated fasting homocysteine levels, and by 1.24-fold in
those with elevated postload homocysteine levels. Only smoking and
hyperhomocysteinemia were strong predictors for coronary heart disease among
our study group.
Conclusion: Hyperhomocysteinemia is significantly and independently
associated with coronary heart disease in our Jordanian sample.
Key
words: Homocysteine (Hcy), Coronary Heart
Disease (CHD), Hyperhomocysteinemia, Methionine Loading Test.
JRMS
June 2004; 11(1): 7-16
Introduction
The major risk factors of coronary heart disease (CHD)
have been identified in both retrospective and prospective studies. However, hyperlipidemia, hypertension,
smoking, diabetes mellitus, obesity, and alcohol intake are all known CHD risk
factors. To date, many studies indicate that hyperhomocysteinemia is a very
well recognized risk factor for CHD (1).
Homocysteine (Hcy), a sulfur- containing amino- acid, is
formed in the metabolic pathway of methionine that is obtained from either
plant or animal origin proteins in the diet. It is also referred to as a
metabolite (2)
or as an
intermediate of methionine (3). In humans, homocysteine is remethylated to
methionine by folate and cobalamine dependent enzymes, and catabolized to
cysteine by vitamin B6 dependent enzyme. Thus, any disturbance in the metabolic
pathway of methionine, due to genetic defect and/or nutrient deficiencies,
leads to Hcy accumulation in the circulation (4).
Many studies have
shown that moderate hyperhomocysteinemia is associated with increased risk of
coronary atherosclerosis, thrombosis, cerebrovascular disease, and peripheral
vascular disease. Verhoef et al.
reported that elevated plasma Hcy is an independent risk factor for myocardial
infarction (5).
Hyperhomocysteinemia can be caused by other environmental
determinants that may be more frequent than some genetic factors (6). These determinants include nutrient
deficiencies of folate, vitamin B6, and vitamin B12,
alcohol intake, smoking, chronic diseases, increasing age and being a male (7).
The role of moderate hyperhomocysteinemia as an
independent risk factor for CHD has been recently supported in many
case-control studies (8).
Information on nutrition and health status of the Jordanian population
is relatively scanty.
To date, only one study has been conducted to investigate
hyperhomocysteinemia as a risk factor for CHD in an Arab population (9). However, it has been revealed that elevated
level of Hcy is an independent risk factor for CHD among Arab men in Syria (9). Furthermore, the identification of all
possible risk factors for CHD is critical in disease prevention and therapy in
high-risk populations.
The information obtained from the present study will help
in evaluating the role of hyperhomocysteinemia in CHD mortality and
morbidity. In addition, results from
this study will provide a baseline data for further studies.
Finally, the knowledge gained from this study will be
helpful in CHD prevention and therapy, particularly, in high-risk individuals.
The purpose of this study was to investigate the relation
between tHcy and CHD risk in Jordanian subjects, as well as the relation
between hyperhomocysteinemia and other conventional risk factors.
Methods
Subjects and Data Collection
In the present case-control study, 45 patients known to
have CHD of either sex and 35 healthy controls were examined. All subjects were recruited from King Hussein
Medical Center in Amman, Jordan during the whole year 1999. The protocol approval was obtained from both
Howard University Institutional Review Board (IRB) and the Protection of Human
Subjects Ethics Committee in Jordan. Patients who were diagnosed during the
previous year and confirmed to have clinical evidence of CHD were considered
eligible to participate in the study.
Patients with a history of chronic diseases such as: renal disease,
thyroid disease, alcoholism, diabetes mellitus, cancer, and/or patients on
medications that are known to interfere with total homocysteine (tHcy) metabolism; and pregnant women were
excluded from the study. Systematic
sampling was used for the selection of patients. The controls were selected
from either healthy personnel in the center or their relatives. All participants in the present study were
Jordanians. Informed written consent was
obtained from all subjects who agreed to participate in the study. Data were collected through blood samples and
a questionnaire. Bostom et al.
indicate that measurements of plasma fasting tHcy alone do not detect cases
that might have post methionine load (PML) hyperhomocysteinemia. Hence, in the present study, blood samples
were obtained from all subjects at fasting (before a methionine load) and 4
hours after a standardized methionine load (10).
Three conventional risk factors were examined in the
present study including smoking, hypertension, and hypercholesterolemia. Smoking habit was determined at the time of
CHD diagnosis for patients and at the time of methionine loading test for the
controls. Blood pressure readings were
taken for all subjects before and after the methionine-loading test; and the
average of four readings was taken.
Information on the usage of hypertensive drugs was obtained from either
the medical records (patients) or directly from the subjects (controls). Cholesterol levels for the patients were
obtained from the medical records. Blood
samples were obtained from the controls at fasting and were analyzed for serum
cholesterol levels on the first day of participation.
The questionnaire provided information on age, gender,
ethnic group, height, weight, smoking, and vitamin intake. The form was completed on the first day of
participation.
Definition of Variables
Age was analyzed as both a continuous and a categorical
variable. Plasma tHcy level above the 75th percentile of the
controls distribution (11) was regarded as
hyperhomocysteinemia 12.3 (μmol/L) for fasting tHcy, and 33.6 (μmol/L) for
postload tHcy. Controls with tHcy levels
below the 75th percentile of the controls distribution were used as
reference in odds ratio estimates.
Cholesterol levels were analyzed as either a continuous or a categorical
variable. Cholesterol levels of > 200
mg/dL were considered as hypercholesterolemia.
Subjects with cholesterol levels < 200 mg/dL were used as reference
in odds ratio estimates. Since
cholesterol distribution was similar among cases and controls, the 75th
percentile of controls distribution was also used as a cutoff point (> 220
mg/dL). The odds ratio estimates for
smoking were based on a comparison between current smokers of 20 cigarettes per
day and nonsmokers. Smoking was also
analyzed as a categorical variable in which current smokers were compared to
ex-smokers (who had quit smoking for at least six months before participating
in the study) and nonsmokers.
Hypertension was considered present if the systolic blood pressure was
(160 mm Hg, diastolic blood pressure (95 mm Hg, or if the subject was taking
antihypertensive medication (1). According to the previous definition, only
two subjects had high blood pressure in which both of them were on
antihypertensive drugs. Therefore, the
odds ratio estimates for hypertension were based on a comparison between
subjects taking medication and those who were not (Yes/No) as a reference.
Biochemical Measurement
High performance liquid chromatography (HPLC) with
fluorescence detection was used to determine plasma tHcy levels. Sample
preparation was performed as described by Araki and Sako (12). Since the isocratic HPLC system was more
feasible in the laboratory, the mobile phase was performed as described by
Accinni et al (13).
All prepared blood samples for tHcy analysis were transferred to the
laboratories of Jordan University of Science and Technology (JUST), Irbid,
Jordan.
Statistical Analysis
Mean values and standard deviation of the risk factors
for CHD were determined. Case-control differences were examined using analysis
of variance (ANOVA) and t-test for continuous variables, and chi square for
categorical variables. To examine the
association between tHcy levels and other risk factors, both bivariate and
multivariate analyses were performed.
Odds ratio estimates of fasting and postload tHcy levels were performed
by logistic regression analyses. The
odds ratio estimates plus 95% confidence intervals (CI) are presented for
tHcy levels as a continuous variable (14). The odds ratio was also estimated for tHcy as
a categorical variable defined by the 75th percentile of controls
distribution. Controls with tHcy
levels below the 75th percentile of the controls distribution were
used as reference. To determine whether
tHcy concentrations interact with other conventional risk factors, multiple
logistic regression models were performed.
Two-tailed p values of 0.05 were considered significant. The statistical analyses were performed using
the SPSS Graduate Pack 9.0 for Windows.
Results
Characteristics of the
Subjects
The
major characteristics of the patients and the control subjects are presented in
Table I. Age and gender distribution did
not differ between patients with CHD and the controls. The prevalence of
hypertension, cholesterol levels, and smoking habits were all similar among
cases and controls. The mean level for systolic blood pressure was not
significantly higher in controls than in cases (p=0.06). The prevalence of hypertension was 29% in
patients and 31% in controls. Among
cases, 38% of the patients had elevated cholesterol levels (> 200 mg/dL) as
compared to 31% of the controls.
Although 60% of the patients were current smokers versus 46% of the
controls, the difference between the two groups was not significant (p= 0.08).
Pearson correlation tests showed that age was directly and significantly
associated with smoking (r =0.3, p= 0.008) and systolic blood pressure (r =
0.27, p= 0.01). Systolic blood pressure
was positively associated with cholesterol levels (r = 0.24, p= 0.02) and body
mass index (BMI) (r = 0.23, p= 0.04).
The adjustment for age and gender did not affect the associations.
The distribution of fasting and postload tHcy levels was
shifted toward higher values in patients as compared to controls. Postload tHcy levels were positively and
significantly associated with fasting tHcy levels (r=0.73, p= 0.00) and the
increase in tHcy as well (r=0 .8, p= 0.04).
The mean fasting and postload tHcy levels were 29% and
16%, respectively, higher in patients than in controls (p<0. 05). The total
increase in tHcy was also examined (postload minus fasting). The increase in tHcy did not differ among
the subjects.
A total of 60% of cases had hyperhomocysteinemia as
compared to 40% of controls. Postload
tHcy levels was identified in 6 (7.5%) more subjects (patients and controls)
who were not classified by fasting tHcy levels.
Similarly, the increase in tHcy was identified in 4 (5%) additional
subjects (patients and controls) who were not identified by either fasting or postload
tHcy levels.
Plasma Homocysteine and
other Risk Factors
The relationship between tHcy concentrations and each
risk factor was examined by bivariate analysis (Table II). Hypertension,
cholesterol levels, and smoking were significantly related to homocysteine
levels (p < 0.05) in patients with low risk profiles. Homocysteine levels did not differ between
males and females. Age was not
significantly related to Hcy levels (p=0.09). Patients with cholesterol levels
less than 200 mg/dL had significantly higher tHcy levels as compared to their
respective controls (p= 0.01).
Similarly, patients who were normotensives (p= 0.01) and nonsmokers
(p=0.02) had significantly higher tHcy levels as compared to controls. Plasma tHcy levels were not significantly
higher among smokers, as compared to controls (p= 0.08). However, when all risk
factors were included in the model, only hypertension and smoking remained
significant (p < 0 .05). No
interaction effect was found between tHcy and the conventional risk factors,
even after controlling for age and gender (age and gender were included in the
model as covariates).
Relative Risks for Elevated
tHcy
The
odds ratios for patients with and without elevated fasting and postload tHcy
levels were determined by single logistic regression analyses. In these analyses, both fasting and postload
levels were significantly related to CHD risk (Table III). Among patients with elevated fasting tHcy levels,
the odds ratio for tHcy levels was 1.85 [(1.3-2.5), p= 0.00] as compared to
subjects below the 75th percentile of the controls
distribution. For postload tHcy levels,
the odds ratio was 1.24 [(1.1-1.4), p= 0.00] as compared to the reference
group.
For patients without fasting and postload elevated
tHcy levels, the odds ratio for tHcy levels was decreased but remained a strong
predictor for CHD (p <0 .05).
The odds ratio for tHcy levels was examined with the
presence of each risk factor separately.
In the presence of all risk factors including
hypertension, hypercholesterolemia and smoking, the odds ratio for patients
with elevated fasting tHcy levels were 3.0 [(1.5-5.9), p=0. 001] as compared to
the reference group; and 1.14 [(1.03-1.26), p= 0.007] for smokers of 20
cigarettes per day or higher as compared to nonsmokers. Adjustment for age and gender did not affect
the significance of the prediction. In
this analysis, hypercholesterolemia, and hypertension were not significantly
related to CHD (Table IV). Since the
results did not show any statistical significance, only the combined, age and
sex-adjusted results are presented.
However, when adjusted for the presence of smoking, the odds ratio for
fasting tHcy levels were reduced but remained significant predictors of CHD
[odds ratio =1.9 (1.3-2.6), p=0 .0001].
Similarly, in patients with elevated postload tHcy, the
odds ratio for tHcy levels was 1.3 [(0.17-3.6), p= 0.0002)] as compared to
subjects with tHcy levels below the 75th percentile of controls
distribution; and 1.0 [(1.0-1.16), p=0.01] for smokers of 20 cigarettes per day
as compared to nonsmokers (Table IV).
The relationship between tHcy levels and the risk for CHD was
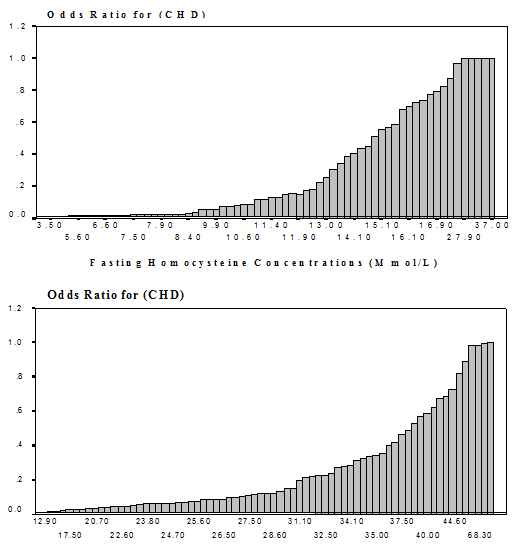
examined. Fig. 1 illustrates the
continuous relationship between fasting and postload tHcy levels with the risk
of CHD in patients as compared to controls.
The estimated odds ratio for CHD per 5-μmol/L increments in tHcy was 2.1
[(1.2-3.8), p=0.0008]. Finally, no
interaction effect was observed between fasting or postload tHcy levels and the
conventional risk factors.
Discussion
Our findings demonstrate the association between tHcy
concentrations and CHD risk among Jordanian subjects. It is believed that
fasting tHcy concentration is an indicator of poor remethylation pathways
resulting from either genetic defects and/or folate and cobalamine deficiencies. The postload methionine level reflects
cystathionine β synthase (CBS) impairments, a vitamin B6
dependent enzyme (6).
A significant increase in either fasting or postload tHcy
ranges 10-30% in patients as compared to controls and
was between 13 case-control
studies (11). The mean fasting tHcy level was 29%
higher in patients than in controls (p=0.003).
Among patients, 44.4% had elevated fasting tHcy levels as compared to
25.7% of controls (p= 0.03). Our results were consistent with the results of
other studies.
Bostom et al indicated that using fasting tHcy
levels alone in determining hyperhomocysteinemia may result in
misclassification of more than 40% of subjects who may have postload
hyperhomocysteinemia (10).
Graham et al reported that a further 27% of patients with
hyperhomocysteinemia were identified by a postload methionine test (1).
In
our study, fasting tHcy levels identified the majority of the
hyperhomocysteinemia patients. Only
three (6.6%) additional patients were identified by postload tHcy
concentrations. Although the mean
increase in tHcy (postload minus fasting) levels did not differ between
patients and controls, approximately 6% of the patients with elevated tHcy
levels who were not identified by either fasting or postload, were identified
by this measure. Fasting and postload
levels were correlated (r=0.7, p=0.00).
The mean for postload tHcy was 16% higher in patients than in controls (p=
0.02) (Table I).
The prevalence estimates of the major risk factors for CHD
in Jordan in 1991 were 32% for hypertension, 46% for hypercholesterolemia, and
48% and 10% for smoking among males and females, respectively (15). In our sample, the prevalence of hypertension
was 30%, 36.2% for hypercholesterolemia, and 54% for smoking. The differences in major risk factors of CHD
between patients and controls were not as expected. The prevalence of hypertension and
hypercholesterolemia did not differ between patients and controls. Smoking habit was non significantly more
prevalent among patients (60%) as compared to controls (46%) (p=0.08).
The association between tHcy levels and other
conventional risk factors were examined.
Age and gender are known to influence tHcy levels. Plasma tHcy levels tend to be higher among
older male subjects. Genders differences may be related to hormonal factors
and/or muscle mass (3).
Our results did not show a significant age-sex relationship with tHcy
concentrations. Plasma tHcy levels were
not significantly higher in patients older than 50 years as compared to their
respective controls.
Plasma tHcy levels increase with age and are higher among
males than females. Due to the fact that
age and gender differences are related to diet and vitamin status (8),
the contradiction in our findings cannot be explained in the absence of vitamin
measures.
Many studies have addressed the association between
hyperhomocysteinemia and the conventional risk factors. Graham et al reported interaction events
between tHcy and two
risk factors (1).
Elevated tHcy levels interacted strongly with both hypertension and
smoking. The joint effect was most
pronounced among women as compared to men.
Results from a recent study showed that fasting tHcy
levels tend to be significantly higher among hypertensive Chinese subjects
(p< 0.05) (16).
Verhoef et al reported a positive correlation
between high blood pressure and tHcy levels in patients with CHD as compared to
controls (17). Nygard
et al illustrated positive correlation between elevated tHcy levels and
other risk factors including age, gender, smoking, hypertension, and
hypercholesterolemia (18).
Glueck et al
reported significant tHcy interaction effects with high-density lipoprotein
(p=0.012) and triglycerides (p=0.02) in atherosclerotic patients with hyperlipidemia
(19). Hoogeveen et
al reported an interaction between hyperhomocysteinemia and type 2 diabetes
to risk of 5-year mortality (20). The odds ratio for hyperhomocysteinemia was
2.5 (1.07-5.91) in diabetics as compared to 1.3 (0.87-2.06) in non diabetics (p
for interaction = 0.08).
In our study, fasting tHcy levels were highly related to
hypertension and smoking, but not hypercholesterolemia (P value <
0.05). In these models, fasting tHcy
levels were significantly higher among normotensive patients as compared to
controls (p= 0.01). Similarly, fasting
tHcy levels were more pronounced among patients with normal cholesterol levels
(< 200 mg/dL) and nonsmokers (p<0 .05) (Table II). Results from a recent study showed that the
association between tHcy levels and MI was stronger among subjects with normal
hypertension as compared to hypertensives (21).
Interestingly,
when these risk factors were examined among patients with elevated fasting tHcy
levels (12.3 (μmol/L)), only the prevalence of smoking was significantly higher
among patients as compared to their respective controls (p=0.01).
Moreover, the association between postload tHcy and other
risk factors was also examined. Among
the risk factors, only smoking was significantly associated with postload tHcy
levels (p= 0.01). The association
between postload tHcy and CHD was independent of smoking. The postload levels were significantly higher
among patients as compared to controls irrespective of their smoking habits
(p< 0.05). Multiple regression
analyses did not show interaction events between hyperhomocysteinemia and other
risk factors.
Lack of association between elevated tHcy levels and both
hypertension and hypercholesterolemia that was observed in this study may be
related to the predominance of patients with normal hypertension and
cholesterol levels, or possibly due to the similarity among patients and
controls in terms of these two risk factors.
In addition, the 1999 WHO criteria for the definition of hypertension
was not used, another text criterion was used instead (1)
which may provide another explanation for the observed association between
hyperhomocysteinemia and hypertension.
Serfontein et al reported a significant
association between decreased vitamin
B6 levels and
smoking (22).
The authors suggested that these results might provide another
explanation by which smoking may be involved as a CHD risk factor. Accordingly, the highly significant association
that was observed in the present study between smoking and postload tHcy level
could be related to vitamin B6 status since postload tHcy
level is an indicator for CBS, a vitamin B6 dependent enzyme.
The odds ratio
of CHD was
estimated for both fasting
and postload tHcy concentrations. The
odds ratio for fasting tHcy levels in patients as compared to controls was 1.2
[(1.04-1.3), p=0 .008] and 1.1 [(1.0-1.11), p=0.03] for postload tHcy
levels. Among patients with elevated
fasting tHcy, the odds ratio was 1.9 [(1.3-2.5), p=0.00]. For elevated postload tHcy, the odds ratio
was lower but remained significant [odds ratio = 1.2 (1.1-1.39), p= 0.00]
(Table III). The observed odds ratio in
our study is consistent with those reported by a recent nationally
representative study of US adults (NHANES III) relating elevated tHcy levels
with MI risk (21). In
this study, the odds ratio for hyperhomocysteinemia in blacks and whites was
1.9 (.8-4.2) and 1.8 (1.1-3.1), respectively.
Although
the observed odds ratio for CHD was lower among patients without elevated tHcy
levels, fasting and postload tHcy levels remained strong predictors for CHD
(Table III). The increase in tHcy
(postload minus fasting) was not related to CHD risk. In the present study, we illustrated a
significant continuous relationship between elevated tHcy levels (fasting and
postload) and CHD risk (Fig. 1). The
increase in the odds ratio becomes more pronounced in the middle of tHcy
distribution. These observations suggest
a dose-response relationship between tHcy levels and CHD risk. The estimated odds ratio for CHD per 5 (μmol/L
increment of fasting tHcy was 2.1
[(1.2-3.8), p= 0.008]; and 1.3
[(1.0-1.7), p=0.03] for postload levels.
Hypertension and hypercholesterolemia were not significantly related to
CHD risk among the study population.
Only smoking and elevated fasting and postload tHcy levels were strong
predictors of CHD. In the presence of
other risk factors, the odds ratio for a case with elevated fasting tHcy (12.3
(μmol/L) was increased
by 1.15, and 0.05 for elevated postload tHcy levels (33.6 (μmol/L). The odds ratio for a smoker of 20 cigarettes
per day with elevated fasting tHcy was (1.0-1.26) (Table IV).
We compared our results with those of other case-control
studies relating elevated tHcy levels to the risk of CHD. A meta analysis showed that the estimated
relative risk (RR) for coronary artery disease per 5 (μmol/L increase in tHcy
levels was 1.6 (1.4-1.7) for
men and 1.8
(1.3-1.9) for women (23).
Chambers et al investigated plasma tHcy levels and
its association with CHD risk among 551 male cases (294 Europeans, 257 Indian-
Asian) (24). The odds
ratio per 5 (μmol/L increment in fasting tHcy was 1.2 (1.0-1.4) for Indian
Asians, and 1.3 (1.1-1.6) in
Europeans. The corresponding
odds ratios for CHD
among European subjects that were previously reported by Graham et al
was 1.3 (1.1-1.6) for men and 1.4 (1.0-2.0) for women (1).
The mechanism by
which hyperhomocysteinemia is
related to CHD
risk is not
fully understood. One
of the clinically
proposed mechanisms is
that it may
affect the cells
through formation of injurious oxygen
species (25). It
may also affect platelet function and the coagulation
system (26).
As in many developing countries, Jordan experienced
trends toward urbanization that is accompanied by changes in life style. According to the Ministry of Agriculture in
Jordan, 60 % of the total energy intake is derived from carbohydrates, 20% from
fat, and 14% from protein (Ministry of Health 1991). Joubran et al indicated that the
consumption of vegetables and fruits among Arabs is relatively limited. We believe that this trend may remain because
the prices of fruits, vegetables, and fortified cereals are high. In our study population, none of the
subjects used vitamin supplementations or was physically active.
Many studies have shown that hyperhomocysteinemia can be
lowered by folic acid administration and additional improvement has been
achieved when vitamin B12 was added. Information on both dietary intake and
biochemical status of B vitamins among Jordanians is not available. Therefore,
efforts should be made to satisfy the needs in this research area. In a recent prospective, double-blinded,
randomized trial that involved 205 patients who had successful coronary
angioplasty, Schnyder et al examined the effect of Hcy lowering with a
combination of folate, vitamin B6, and vitamin B12
on restenosis. The subjects were
divided into 2 groups to which they were randomly assigned to receive either
folate treatment (1 mg folic acid, 400µg vitamin B12, 10 mg
pyridoxine) or a placebo. The results
showed significant reductions in plasma Hcy levels and in the rate of
restenosis after coronary angioplasty (P < 0.00). The authors suggested that lowering Hcy
levels by folic acid supplementation should be considered in the treatment of
patients undergoing coronary angioplasty (27). Due to the observed similarities with regards
to the prevalence of risk
factors for CHD
among our study
population, it is suggested
that screening for
controls should be
more intensive and rigorous. Finally, based on our observations, we
suggest that routine screening for hyperhomocysteinemia should be considered
particularly among patients with high-risk profile.
Conclusion
The present study illustrates for the first time that
hyperhomocysteinemia is significantly and independently associated with
coronary heart disease in our Jordanian sample.
Limitations of the
Study
The present study had some limitations. Homocysteine levels are inversely related to
B vitamins including folate, cobalamine, and vitamin B6.
Information on dietary intakes and serum levels of these vitamins among the
study population was not available.
Hence, the extent of the relationship between tHcy levels and B vitamins
as risk factors for CHD could not be fully explored. Second, the number of subjects that were
examined as compared to those in other case-control studies was relatively
small. Methionine has an unpleasant
taste and methionine loading is difficult to apply, therefore, recruitment of
subjects was not feasible. Matching the
cases with subjects from the personnel was relatively difficult due to
demographic variations between both groups.
Subjects from the personnel or their relatives were younger and mostly
males, which made the matching more complicated. Therefore, recruitment of controls was
performed without matching with cases of CHD.
However, in spite of the relatively small sample size, the strength of
the association between tHcy levels and CHD risk that were obtained by many
statistical models contributes to the strength of these findings. Finally, the number of controls was lower
than cases.
Table I.
Demographic, clinical, laboratory characteristics among cases and
controls.
Variables
|
Patients (n=45)
|
Controls (n=35)
|
P. Value
|
|
Mean age*, years
|
46.7 ± 8.02
|
47.6 ± 7.7
|
0.6
|
|
Males (%)
|
35 (77.7)
|
25 (71.4)
|
0.6
|
|
Females (%)
|
10 (22.2)
|
10 (28.5)
|
0.6
|
|
Body Mass Index*, Kg/m2
|
29.6 ± 7.7
|
27.4 ± 4.6
|
0.1
|
|
Systolic Blood Pressure, mm Hg*
|
112 ± 16.46
|
118 ± 16.5
|
0.06
|
|
Diastolic Blood Pressure, mm Hg*
|
71.33 ± 11
|
74.43 ± 8.64
|
0.1
|
|
No. (%) with Hypertension †
|
13 (28.8)
|
11 (31.4)
|
0.81
|
|
Mean Cholesterol Levels, mg/dL*
|
191.13 ± 47.0
|
181± 45.0
|
0.34
|
|
No. (%) of Current Smokers
|
27 (60)
|
16
(45.7)
|
0.08
|
|
Mean Cigarettes/day
|
14.93 ± 13.6
|
10.6 ± 12.3
|
0.14
|
|
Mean Fasting Homocysteine, μmol/L*
|
14.12 ± 7.39
|
10.0 ± 3.8
|
0.003
|
|
Mean Postload Homocysteine, μmol/L*
|
32.86 ± 12.37
|
27.6 ± 7.12
|
0.02
|
|
Total Homocysteine Increase, μmol/L‡
|
18.7 ± 8.6
|
17.7 ± 5.5
|
0.52
|
* Values are expressed
as means ± SD. † Subjects using antihypertensive
drugs. ‡ Postloading minus fasting
total homocysteine.
Table II. Fasting homocysteine concentrations by CHD
risk factors for patients and controls.
|
Homocysteine Concentrations*
(mmol/L)
|
|
Variable
|
No. (%)
|
Patients
|
No. (%)
|
Controls
|
|
Smoking Habit
|
|
|
|
|
|
Nonsmokers†
|
5 (11.1)
|
19.11a ± 8.8
|
13 (37.1)
|
10.45b ± 4.3
|
|
Exsmokers
|
13 (28.8)
|
11.76a ± 7.8
|
6 (17.1)
|
12.8a ± 3.9
|
|
Smokers
|
27 (60)
|
14.65a ± 6.97
|
16 (45.7)
|
8.4a ± 2.6
|
|
Cholesterol (mg/dL)
|
|
|
|
|
|
< 200†
|
30 (66.6)
|
15.08a ± 7.8
|
22 (62.8)
|
9.89b ± 3.9
|
|
³ 200
|
17 (37.7)
|
12.27a ± 5.6
|
11 (31.4)
|
9.5a ± 3.8
|
|
Hypertension
|
13(28.8)
|
11.55a ± 5.9
|
11 (31.4)
|
9.8a ± 3.7
|
|
No Hypertension†
|
32 (71.1)
|
15.16a ± 7.7
|
24 (68.5)
|
10.02b ± 3.9
|
|
Gender
|
|
|
|
|
|
Males
|
35 (77.7)
|
13.9a ± 6.5
|
25 (71.4)
|
10.15a ± 3.7
|
|
Females
|
10 (22.2)
|
14.56a ± 10.5
|
10 (28.5)
|
10.05a ± 4.4
|
|
Age
|
|
|
|
|
|
29-39 years
|
9 (20)
|
14.15a ± 9.3
|
5 (14.2)
|
11.6a ± 3.8
|
|
40-50 years
|
21 (46.6)
|
12.7a ± 5.1
|
17 (48.5)
|
9.5a ± 4.2
|
|
51-60 years
|
15 (33.3)
|
16.0a ± 8.8
|
13 (37.1)
|
9.8a ± 3.2
|
*Values are presented as means ± SD.
†Means in the rows with
unlike superscripts (a,b) are significantly different (p £ 0.05)
Table III. Odds
ratio for CHD in subjects with and without elevated homocysteine*
|
Variable
|
Odds ratio (95% CI)
|
P. Value
|
|
Elevated Homocysteine †
|
|
|
|
Fasting
|
1.9 (1.3-2.5)
|
0.000
|
|
Postload
|
1.2 (1.1-1.39)
|
0.000
|
|
No Elevated Homocysteine †
|
|
|
|
Fasting
|
0.8 (0.72-0.95)
|
0.01
|
|
Postload
|
0.9 (0.84-0.97)
|
0.009
|
*Patients with tHcy levels above and below the 75th percentile of
the controls distribution.
† Reference group: Controls below the 75th percentile
of controls distribution.
Table IV. Odds
ratio for elevated fasting and postload homocysteine with other risk factors
(Adjusted for Age and Gender)
|
Variable
|
Odds ratio (95% CI)
|
P. Value
|
|
Elevated Homocysteine*
|
|
|
|
Fasting
|
3.0 (1.5-5.9)
|
0.001
|
|
Hypercholesterolemia†
|
1.4 (0.2-8.4)
|
0.6
|
|
Hypertension‡
|
0.8 (0.11-5.2)
|
0.8
|
|
Smoking (> 20 Cigarettes/d)§
|
1.1 (1.03-1.26)
|
0.007
|
|
Elevated Homocysteine*
|
|
|
|
Postload
|
1.3 (0.17-3.6)
|
0.0002
|
|
Hypercholesterolemia†
|
0.9 (0.17-3.6)
|
0.7
|
|
Hypertension‡
|
0.6 (0.09-3.4)
|
0.5
|
|
Smoking (> 20 Cigarettes/d) §
|
1.0 (1.0-1.16)
|
0.01
|
*Patients with tHcy
levels above the 75th percentile of the controls distribution as
compared to controls below the 75th percentile of the controls
distribution. †Subjects with cholesterol
levels > 200 mg/dL. ‡Subjects
using antihypertensive drugs.
§Smokers of 20 cigarettes per day or greater as compared to
nonsmokers.
|
|
|
|
|
|
Fig.
1.
Association Between Fasting and Postload Elevated Homocysteine Levels
and Relative Risk for CHD*
*
Mean predicted odd ratios and actual tHcy levels. |
References
1. Graham IM, Daly LE, Refsum HM, et al. Plasma
homocysteine as a risk factor for vascular disease. JAMA (1997); 277:1775-1781.
2. Shils
ME, Olson JA, Shike M, Ross AC. Modern nutrition in health and disease.
Williams & Wilkins Awaverly Company. 1999; 136-139.
3. Nygard O, Nordrehaug JE, Refsum H, et al. Plasma homocysteine levels and
mortality in patients with coronary artery disease. N Engl J Med (1997); 337:230-236.
4. Bendich
A, Deckelbaum RJ. Preventive nutrition.
Homocysteine, folic acid, and cardiovascular disease risk. Human press, Totowa, New Jersey. 1997.
5. Verhoef
P, Stampfer MJ. Prospective Studies of Homocysteine and
Cardiovascular Disease. Nutr Rev 1995; 53(10): 283-288.
6. Sacco
RL, Roberts JK, Jacobs BS.
Homocysteine as a risk factor for ischemic stroke: An epidemiological story in
evolution. Neuroepidemiology 1998; 17: 167-173.
7. Moghadisian
MH, McManus BM, Frohlich JJ. Homocysteine and coronary artery
disease. Arch. Intern Med (1997);
157: 2299-2308.
8. Nygard
O, Volest SE, Refsum H, et al. Total homocysteine and cardiovascular disease. J Intern Med (1999); 246: 425-454.
9. Joubran
R, Asmi M, Busjahn A, et al. Homocysteine levels and coronary heart disease in Syria. J Cardiovasc Risk 1998; 5: 257- 261.
10. Bostom
AG, Jacques PF, Nadeau MR, et al. Post-methionine
load hyperhomocysteinemia in persons with normal fasting total plasma
homocysteine: Initial results from the NHLBI family heart study. Atherosclerosis 1995; 116: 147-151.
11.Christen
WG, Ajani UA, Glynn RJ, Hennekens CH. Blood levels of homocysteine and increased risks of cardiovascular
disease. Arch Intern Med 1995;
160: 422-434.
12. Araki
A, Sako Y. Determination of free and total homocysteine
in human plasma by high-performance liquid chromatography with fluorescence
detection. J
Chromatogr
1987 ; 422: 43-52.
13. Accinni R, Campolo J, Bartesaghi S, et al. High-performance
liquid chromatographic determination of total plasma homocysteine with or
without internal standards. J Chromatogr 1998; 828: 397-400.
14. Stephen
A.
Data Analysis with SPSS. A Viacom
Company. By Allyn and Bacon. Massachusetts, USA. 1999.
15. Ministry
of Health. Ten-year Plan
(1997-2006) for control of Cardiovascular Diseases in Jordan. Amman, Jordan, 1991.
16. Huey-Herng
Sheu W, Wen-Jane Lee, Ying-tsung Chen. Plasma homocysteine concentrations and insulin sensitivity in hypertensive
subjects. Am J Hypertens 2000;
13: 14-20.
17. Verhoef
P, Stampfer MJ, Buring JE, et al. Homocysteine metabolism and risk of myocardial
infarction: Relation with vitamin B6, B12,
and Folate. Am J Epidemiol 1996;
143: 845-859.
18. Nygard
O, Vollset SE, Refsum H, et al. Total plasma homocysteine and cardiovascular risk profile. JAMA (1995), 274: 1526-1533.
19. Glueck CJ, Shaw P, Lang JE, et al. Evidence that homocysteine is an independent risk factor
for atherosclerosis in hyperlipidemic patients.
Am J Cardiol 1995; 75: 132-136.
20. Hoogeveen EK, Kostense PJ, Jakobs C, et al. Hyperhomocysteinemia increases risk of death, especially
in type 2 diabetes: 5-year follow-up of the hoorn study. Circulation 2000; 101: 1506-1511.
21.Giles
WH, Croft JB, Greenlund KJ, et al. Association
between total homocystine and the likelihood for a history of acute myocardial
infarction by race and ethnicity: Results from the third national health and
nutrition examination survey. Am Heart J 2000; 139: 446-453.
22. Serfontein
WJ, Ubbink JB, De Villiers LS, Becker PJ. Depressed plasma pyridoxal-5-phosphate levels in
tobacco-smoking men. Atherosclerosis 1986; 59: 341-346.
23. Boushey
CJ, Beresford SAA, Omenn GS, et al. Assessment of plasma homocysteine as a risk factor for
vascular disease: Probable benefits of
increasing folic acid intakes. JAMA 1995; 274: 1049-1056.
24. Chambers
JC, Obeid OA, Refsum H.
Plasma homocysteine concentrations, and risk of coronary heart disease in UK
Indian Asian and European men. Lancet
2000; 355: 523-27.
25. Jacobsen
DW. Homocysteine and vitamins in
cardiovascular disease. Clin Chem
1998; 44:8(B) 1833-1843.
26. Mayer
EL, Jacobsen DW, Robinson K. Homocysteine and coronary atherosclerosis. JACC
1996; 27(3): 517-527.
27. Schnyder G, Roffi M, Pin R, et al. Decreased Rate of Coronary Restenosis after Lowering of
Plasma Homocysteine Levels. N Engl J
Med 2001; 345(22): 1593-1600.