Abstract
Objectives: To
study whether advanced maternal age and maternal grandmother age are associated
with increased risk of Down syndrome siblings in a group of Jordanian families.
Methods: This study was conducted on 127 confirmed Down
syndrome cases with the age range of 18 weeks gestation to 15 years old, which were
referred between the period of 2005-2008 for cytogenetic analysis at the
Cytogentics section, Princess Iman Research and Laboratory Sciences Center/King
Hussein Medical Center. Maternal and grandmaternal mother ages were obtained
directly from the study group when the samples were collected from siblings.
The maternal age ranged between 19-45 years while the maternal grandmother’s
age ranged between 15-49 years. One
hundred healthy families were randomly recruited from the hospital staff as a
control group. Logistic regression was used for statistical analysis.
Results: One
hundred seventeen down syndrome cases had
free trisomy 21, 7 with translocation, 2 mosaic and one with double aneuploidy (47,XXY,
+21). Fifteen cases were diagnosed prenatally while 112 were diagnosed
postnatally. The effect of maternal age and maternal grandmother age were found
to be significant using logistic regression statistics (P = 0.001; OR=
2.816; 95% CI, 1.48-5.33) for the mother’s age and (P = 0.001; OR=
2.902; 95%CI, 1.521-5.53) for the grandmother’s age.
Conclusion: Advanced maternal and maternal grandmother ages are
risk factors for Down syndrome. More studies and investigations are needed for
better understanding of the biological factors responsible for the proper
meiotic segregation of germ cells during the fetal development of the embryo in
advanced maternal and grandmother’s age.
Key word: Advanced
grand maternal age, Chromosomal aneuploidy, Down syndrome, Robertsonian
translocation
JRMS September 2010; 17(3): 51-56
Introduction
Around 50% of spontaneous abortions before 15
weeks of gestation are chromosomally aneuploid, with trisomies accounting for
50% of abnormal abortions.(1) Trisomy 21,
the chromosomal abnormality responsible for >95% of individuals with Down
Syndrome (DS), is the most commonly identified cause of mental retardation,
with an incidence of 1 in 600 live births.(2) While the incidence of fetal trisomies is
directly related to advanced maternal age,
no specific genetic factors had been identified thus far.(3) The mechanisms for maternal meiotic
non-disjunctional events are under study. Generally, children with Down
syndrome have “free” trisomy 21 (92-95%), mosaic trisomy 21 (2-4%), or trisomy
21 due to a Robertsonian translocation (3-4%).(4)
An association has been
found between the risk of Down syndrome and the age of the maternal grandmother at the
mother’s birth.(5-7) Female meiosis starts in fetal life, and
nondisjunction in the first meiotic division of a female might be induced
during the fetal period, especially if her mother is older.
At least in
5% of all clinically recognized human pregnancies, meiotic segregation errors
give rise to zygotes with the wrong number of chromosomes. The nondisjunction
error is more frequent in first meiotic division (80%) rather than second
meiotic division (20%).(8) The
polymorphic microsatellites have revealed that Trisomy 21 is due to
nondisjunction of 90% of the maternal and 10% of paternal chromosome.(9) These observations have led to the hypothesis
that chromosome 21 nondisjunction requires two hits: The first hit, which
occurs during fetal meiosis, establishes bivalents with ‘susceptible’ meiotic
configurations. The second hit involves an age-related degradation of a meiotic
process which increases the risk of improper segregation for these susceptible
bivalents. Under normal meiotic conditions, the presence of a single chiasma-regardless
of its location is sufficient for proper chromosome segregation. However, as
the ovary ages, a decay or breakdown in the meiotic apparatus (e.g. a spindle
component or sister chromatid cohesion protein) may occur, disturbing the
meiotic process. At this point, certain exchange configurations may be more
likely to undergo improper segregation and non-disjunction. In this manner, as
the age of a woman increases, so too does her chance of a meiotic disturbance.(10,11)
This study was conducted to
assess whether maternal and /or maternal grandmother’s age is associated with
increased risk of Down syndrome siblings in a group of Jordanian families.
Methods
A total of 127 confirmed
DS cases with the age range of 18 weeks (prenatal)
gestation to 15 (postnatal) years, were referred between the period of 2005-2008
for cytogenetic analysis at Cytogenetics section, Princess Iman Research
and Laboratory Sciences Center/King Hussein Medical Center to confirm the
clinical diagnosis of Down Syndrome postnatally or referred with abnormal
ultrasound findings prenatally. (Table I and Table II)
One hundred healthy families
were randomly recruited from the hospital staff as a control group (Table I).
Statistical
analysis with logistic regression was
performed using the SPSS version 10 to record the effect of the variables (Table
III).
Lab procedures
Chromosomal preparations
obtained from phytohemagglutinin (PHA)-stimulated peripheral blood cultures and
Amniomax (Gibco-USA) special media was used for culturing the amniotic fluid
and chorionic villi. All samples were subjected to Giemsa Trypsin Gurr (GTG)
banding and karyotype analysis according to ISCN 1995.
Fluorescence-in-situ Hybridization (FISH) was
performed using AneuVysion Assay Kit (Abbot-Vysis,
USA). Two sets
of combination probes (chromosomes 13, 21 and 18, X, and Y) were used..
Results
One hundred seventeen
DS were found as free trisomy 21, 7 with
translocation (Table IV), two mosaic,
and one with non-classical type (47,XXY, +21). Fifteen cases were diagnosed prenatally and
112 postnatally.
Figure 1 shows the Pedigree of four
families out of the seven families, whom were found to have translocations. Two
families were found to have inherited pattern, one family had t (14; 21) and
the other family diagnosed prenatally had t (13; 21). The paternal karyotype
was normal in five cases suggesting de-novo origin, interestingly one family
has two Children with DS and the parental karyotype was normal. Both mosaic cases were diagnosed prenatally.
Table III presents the
logistic regression of this study. Logistic
regression indicated that the mothers (58%) had advanced age during their
conception of their DS siblings, and grandmothers (60%) had advanced age during
conception of their daughters who gave birth to a DS child.
The effect of maternal age and maternal grandmother
age was found significant, where the logistic regression statistics were P=0.001;
OR= 2.816; 95%CI, 1.48-5.33 for the mother’s age and P=0.001; OR=2.902; 95%CI,
1.521-5.53 for grandmother’s age.
Table I. Study and control age groups
|
Control group age
|
Study group
Age
|
Category
|
|
18
|
19
|
Min maternal's age
|
|
46
|
45
|
Max maternal 's age
|
|
17
|
15
|
Min grandmother's age
|
|
45
|
49
|
Max grandmother's age
|
|
-
|
18wks gestation-15years
|
DS Patients Age range
|
|
32
|
32
|
Average maternal age
|
|
29
|
31
|
Average grandmother’s age
|
Table II. Study group numbers and
their corresponding Down syndrome subtypes
|
Category
|
No
|
%
|
|
Postnatal diagnosis
|
112
|
88
|
|
Prenatal diagnosis
|
15
|
12
|
|
Male
|
76
|
59.8
|
|
Female
|
51
|
40.2
|
|
Translocations
|
7
|
5.5
|
|
Mosaic
|
2
|
1.57
|
|
Free
|
117
|
92.1
|
|
Double aneuploidy 46, XXY, +21
|
1
|
0.78
|
Table III. Logistic regression for the
mother's age and grandmother's age
|
|
OR
Odd ratios
|
95% Confidence interval
|
P value
|
|
Mother's age
|
2.816
|
1.48-5.33
|
0.001
|
|
Grandmother's age
|
2.902
|
1.521-5.53
|
0.001
|
Table IV. Karyotype analysis of seven families with Down syndrome cases with
Robertsonian translocation
|
Karyotype translocations
|
Family Member
|
Mother age
|
Grandmother
age
|
|
Family 1 (C)
|
|
|
|
|
45,XX,rob(13;21)(q10;q10)
|
mother
|
|
|
|
46,XX,rob(13;21)(q10;q10)+21
|
sibling
|
23
|
34
|
|
Family 2 (B)
|
|
|
|
|
46,XX,rob(21;21)(q10;q10)+21
|
sibling
|
23
|
28
|
|
47,XY,+21
|
sibling
|
|
|
|
Family 3 (A)
|
|
|
|
|
45,XY,rob(13;21)(q10;q10)
|
father
|
|
|
|
45,XY,rob(13;21)(q10;q10)
|
sibling
|
30
|
32
|
|
46,XY,rob(13;21)(q10;q10)+21
|
sibling
|
22
|
32
|
|
45,XY,rob(13;21)(q10;q10)
|
father
|
|
|
|
46,XY,rob(13;21)(q10;q10)+21
|
sibling
|
33
|
38
|
|
Family 4(D)
|
|
|
|
|
46,XX,rob(14;21)(q10;q10)+211
|
sibling
|
30
|
35
|
|
Family 5
|
|
|
|
|
46,XX,rob(21,21)(q10;q10)+21
|
sibling
|
25
|
38
|
|
Family 6
|
|
|
|
|
46,XX,rob(21,21)(q10;q10)+21
|
sibling
|
23
|
28
|
|
Family 7
|
|
|
|
|
46,XX,rob(14,21)(q10;q10)+21
|
sibling
|
35
|
40
|
Discussion
Although the effect of maternal age as a risk factor
for Down syndrome (DS) is well known, there are very few reports indicating the
influence of grandmaternal age, on the risk of
their grandchild being born with DS.(12,13) An interesting finding in our study, is that out
of the 127 DS cases studied, only 18 (14.1%) cases have both
mother
and grandmother age < 30 years old,
where the other 109 (85.9%) cases either
or both mother and grandmother
have advanced age during conception of their DS sibling or daughters, respectively.
As shown in the logistic regression (Table III), the present
study demonstrates both maternal and maternal grandmother advanced age (>30)
as risk factors for Down syndrome. If we compare this study with Suttur et
al.’s study,(14) where
logistic regression analysis using the four covariates of maternal age,
grandmother age, father age, and consanguineous marriages together showed that
the effect of maternal age, father's age and consanguineous marriage were diluted
but still of clinical relevance, albeit not statistically significant. However,
the effect of age of the maternal grandmother was not diluted, showing an
increase in odds by 30% per extra year. In our study we found both maternal age
and grandmother advanced age to be significant.
Looking at family pedigrees in his study, it is clear that whenever the
daughter was born to aged mother the chance of this daughter giving birth to DS
children is increased.
How the advanced age of grandmother is responsible
to bring disturbance in the meiosis of her daughter when the grandmother
conceived is explained by Antonarakis.(15) At the
advanced age, the grandmother's reproductive system may fail to produce the
essential proteins like spindle associated proteins, factors responsible for
resting of oocyte, chiasma-binding proteins, DNA repair enzymes, etc. which are
needed for proper meiotic segregation in the germ cells of her daughter. The
non-availability or non-functioning of proteins leads to impairment in the meiotic
process, which in turn results in nondisjunction of chromosome 21 in the oocyte
of the daughter. This event takes place during the embryogenesis of the mothers
of the DS children when she was in grandmother's womb. It is also possible that
recombination is reduced in the oocytes, which brings about the nondisjunction
of chromosome 21. Therefore, DS not only depends on the maternal age but also
on the age of the maternal grandmother which results in nondisjunction of
chromosome 21.(16)
The frequency of
free trisomy 21 observed in this study is most common and is seen in 93%
of cases, where 4.7% had translocation,
1.5% had mosaic, and non-classical (47,XXY, +21) in 0.75%.(17) All these figures are close to those figures
reported in the literature.(4)
Robertsonian
Translocation (RT)
In the present study seven cases were found with
translocation, we screened some members of the families while others we were
unable to contact (Table IV). RT is the
second most common translocation and comprises 4.7% of the cases. Familial
inheritance in Robertsonian
translocation is seen in one
quarter whereas in the remaining it is
de-novo.(18) In this study two families (Fig. 1 A, C) out
of seven were found with inherited
translocation t(13;21). One showed maternal
inheritance (C) and the other one paternal inheritance (A), in the latter, the
mother underwent amniocenthesis and the fetus had balanced translocation, and
after studying the family, we found that his brother is DS case where the
father had unbalanced translocation which he transmitted to the offspring. The
recurrence risk is <1% if the translocation is de-novo. In case of familial RT DS, the genetic risk for female
carrier to have a live born child with translocation DS is about 10%, which
increases to 15% at amniocentesis. For male carriers the recurrence risk to
have a child with translocation DS is about 1%.(19)
Mosaicism
In the
present study two cases (1.5%) were
diagnosed prenatally with mosaicism. Both grandmothers were 40, and 45 years
old respectively and both mothers were young. This figure is similar to that
reported in the literature.(4) Mosaicism
arises after the egg and sperm have fused at conception. As the cells divide
and multiply by ordinary cell division, a chromosome goes astray and a single
cell with an extra chromosome 21 is formed. This cell continues to divide by
ordinary cell division together with the non-trisomic cells and a mixture is
produced.(20)
As with
the other two types of Down's syndrome (apart from when a parent is a carrier)
there is no known reason why mosaic Down's syndrome occurs. It happens equally
often in parents of all ages.
In the
present study, one case (0.78%) had non-classical DS karyotype. These cases
have been reported in major DS studies with a frequency ranging from 0-1.2% and
our figure is consistent with those studies.(21,22)
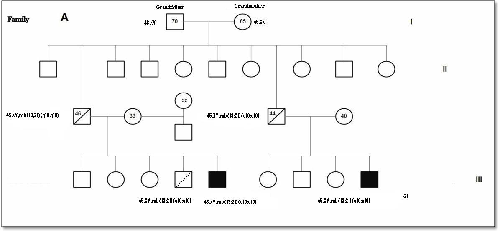
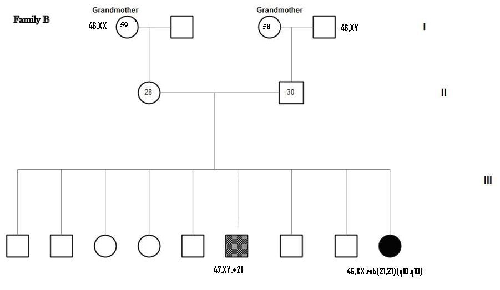
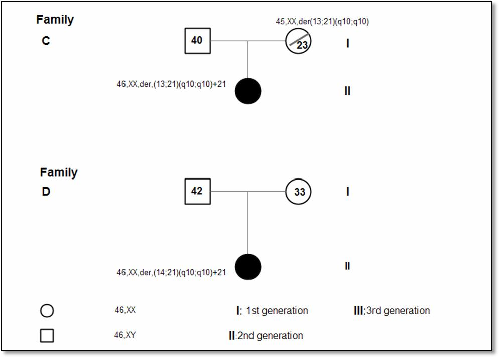
Fig. 1. Pedigrees of 4 families with Robertsonian Translocation (RT)
(Families A and C as shown, have inherited pattern)
predisposition, such factors may play a more important role in the
etiology of the most common double anioploidy 48, XXY, +21, as evident in
the present case (mother’s age was 43 years).(23) The risk of having a child with Down syndrome
increases in a linear fashion until about age 30 and then increases
exponentially thereafter.(4)
Conclusion
Advanced maternal and maternal grandmother ages are
risk factors for Down syndrome. More studies and investigations are needed for
better understanding of the factors responsible for the proper meiotic segregation of germ cells during the
development of the embryo in advanced
maternal and grandmother’s age.
Aknowledgement
Our sincere
thanks to all the staff of cytogenetics section at Princcess
Iman Center/
King Hussein Medical
Center, for compilation
of the data which made this study possible, we would like also to thank Dr.
Yasin Tawarah for the statistical assistance.
References
1. Hassold TJ, Chen N, Funkhouser J, et al. A cytogenetic study of repeated spontaneous abortions. Ann Hum Genent 1980.;44:151-178
2. Fryns, JP. Chromosomal anomalies and autosomal syndromes. Birth defects 1987;23:7-32
3. Hook EB. Rates of chromosome abnormalities at different maternal ages. Obstet Gynecol 1981; 58:282-5
4. Stoll C, Alembik Y, Dott B, Roth MP. Study of DS in 238942 consecutive births. Ann Genet 1998; 41:44–51.
5. Rao, VB. Mean maternal age of Down's syndrome in Hyderabad, India. J Indian Med Assoc 1999; 97:25.
6. Aagesen L, Grinsted J, Mikkelsen M. Advanced grandmaternal age on the mother's side-a risk of giving rise to trisomy 21. Ann hum Genet. 1984; 48:297–302.
7. Papp Z, Varadi E, Szabo Z. Grandmaternal age at birth of parents of children with trisomy. Hum Genet. 1977; 39:221–224.
8. Hassold T, Chiu D, Yamane JA. Parental origin of autosomal trisomies. Ann Hum Genet 1984 May; 48(Pt 2):129-44.
9. Ghosh S, Dey SK. DNA diagnosis of DS using polymerase chain reaction and polymorphic microsatellite markers. Int J Hum Genetics 2000; 10:17–20.
10. Hawley RS, Frazier J, Rasooly R. Separation anxiety: the biology of non-disjunction in flies and people. Hum Mol Genet 1994; 31521–1528
11. Lamb NE, Freeman SB, Savage-Austin A, et al. Non-disjunction of chromosome 21: evidence for initiation of all maternal errors during meiosis I. Nature Genet 1996; 14: 400–405.
12. Lopez PM, Stone D, Gilmour H. Epidemiology of DS in a Scottish city. Paediatr Pinant Epidemiol 1995; 9:331-340
13. Johnson Z, Lillis D, Delany V, et al. The epidemiology of Down syndrome in four counties in Ireland 1981–1990. J Public Health Med 1996;18:78–86
14. Suttur S, Nallur BR. Influence of advanced age of maternal grandmothers on Down syndrome. BMC Med Gene 2006; 7: 4.
15. Antonarakis SE. Parental origin of the extra chromosome in trisomy 21 as indicated by analysis of DNA polymorphisms. Down syndrome collaborative group. N Eng J Med 1991; 324:872-876.
16. Sherman SL, Takaesu N, Freeman SB, et al. Trisomy 21: association between reduced recombination and nondisjunction. Am J Hum Genet 1991;49, 608-620
17. Eid SS, Shawabkeh MM, Hawamdah AA, Kamal RK. Double trisomy 48, XXY,+21 in a child with phenotypic features of Down Syndrome. LabMed 2008; 40: 215-218.
18. Shaffer LG, Jackson-Cook CK, Stasiowski BA, et al. Parental origin determination in thirty de novo Robertsonian translocations. Am J Med Genet 1992;43:957-963
19. Gardner RJM, Sutherland G. Chromosome abnormalities and genetic counseling. J Ment Defic Res., 2nd ed. Oxford, oxford university press 1996; 243-258.
20. Richards BW. Mosaic mongolism J Ment Defic Res 1969; 13(1):66-83.
21. Mokhtar MM, Abd El Aziz AM, Nazmy NA, et al. Cytogenetic profile of Down syndrome in Alexandria. Egypt. East Mediterr Health 2003; 9(1-2):37-44
22. Mutton D, Alberman E, Hook EB. Cytogenetic and epidemiological findings in Down syndrome. England and Wales 1989 to 1993. National Down syndrome Cytogenetic Registry and the Association of clinical Cytogeneticists. J Med Genet 1996; 33:387-394.
23. Kovaleva NV, Mutton DE. Epidemiology of double aneuploidies involving chromosome 21 and the sex chromosomes. Am J Med Genet A 2005; 134:24-32.