ABSTRACT
Objective: To
evaluate the clinical outcome of
different surgical techniques
used in the treatment of giant cell tumor of long bones and their effect on the
rate of local recurrence.
Methods: Thirty-seven patients with giant cell tumor of the long bones have been
treated between July 1994 and July 2003.
All patients were evaluated by clinical examination, plain X-ray, computerized
axial tomography and magnetic resonance imaging. Biopsy was taken in all cases
to confirm the diagnosis and to define the histological grade of the tumor.
Thirty- one patients were treated primarily by curettage and six were treated
primarily by wide excision. Selection of
the surgical technique was based on site and size of the lesion, soft tissue
involvement (intra- or extra-compartmental), tumor grade (histological and
radiological) and if recurrent or not. Patients were followed-up for a minimum
of two years.
Results: The mean age of our patients
at presentation was 29.3 years (ranged from 19 to 52 years) and at last follow
up visit 32.1 years (range 24 to 55 years). Seventeen patients were males and
twenty were females, (male to female ratio was (1:1.2). According to the classification
of Campanacci et al (3 patients were grade I, 24 patients were grade II
and 10 were grade III). There were no mortalities among our cases. Local
recurrence occurred in 9 out of 31 patients treated by curettage.
Conclusion: The main primary treatment of giant cell tumor is
surgery. The use of local adjuvant therapy as part of treatment of giant cell
tumor helps in decreasing the rate of recurrence. Curettage must be extensive
to be effective and requires a large cortical window. Wide excision is used in
extremely large lesions with cortical bone breakthrough and extension into soft
tissue, when the joint could not be preserved and in cases where resection
results in no significant morbidity.
Key words: Adjuvant therapy, Bone cement,
Curettage, Giant cell bone tumor, Reconstruction, Resection.
JRMS
April 2008; 15(1): 23-30
Introduction
The
histogenesis of giant cell tumor (GCT) of the bone is still uncertain. Its
histolopathology does not predict the clinical outcome. Many controversies are
still present regarding the best method of treatment. Giant cell tumor (GCT) of the bone has been
classified by the World Health Organisation (WHO) as "an aggressive,
potentially malignant lesion".
Almost 75%-80% of GCTs have a benign course. The rate of local
recurrence can range from 20% to 50% according to the method of treatment.
Malignant transformation at recurrence is not uncommon and reported to happen
in about 10% of cases. Approximately 1-4% of giant cell tumors give pulmonary
metastases even in cases of benign histopathology.(1-4)
The
most accepted hypothesis regarding the pathogenesis of GCT and its neoplastic
character is the mononuclear spindle-shaped (fibro-osteoblast like) stromal
cells. These cells are genetically unstable, and by secreting a lot of
cytokines and other factors they stimulate the immigration of blood monocytes
into the tumor tissue and promote the formation of the osteoclast - like giant
cells. The characteristic cell types, the monocyes and the giant cells, are
therefore simply reactive components of GCT, while the spindle shaped stromal
cells represent the neoplastic component of the tumor.(5)
GCTs
represent 5% of all primary bone tumors and 21% of all benign bone tumors. It
has a slight female predominance (1.2 -1.5: 1), and occurs most commonly in the
third and fourth decades of life.(6-8) Less than 5% occur in
skeletally immature patients.(9) Approximately 50% of
GCTs are located about the knee at the distal femur and proximal tibia. Most
GCTs are solitary lesions (less than 1% are multicentric).(1,6-8)
Pain is the most common presenting symptom, while swelling and deformity are
associated with large lesions. The incidence of pathological fracture at
presentation is 10-30%.(6-8, 10)
The
diagnosis of GCT of bones depends mainly on clinical and radiological
examination (plain x-ray, computerized tomography and magnetic resonance
imaging) of the site of the lesion. Preoperative biopsy should be taken to
confirm the diagnosis and to define the grade of the malignancy.(1,11,12)
The
grading system adopted by Campanacci et al(7) classifies patients with GCTs, according to
their radiological findings, into grade
I: means no cortical thinning; Grade II: means some cortical thinning and
erosion but no breakthrough; and grade III: means breakingthrough and extension
into the soft tissue.
The
staging system adopted by Enneking in 1986(12) is also used
to stage patients with GCT. It depends on the tumor grade (G), location of the tumor:
intra-compartmental or extra- compartmental (T) and presence or absence of metastasis
(T).
Surgery
is the main treatment of GCTs of long bones with different modalities have been
investigated and used.(6-8, 11, 13-18)
GCT
is not radio-resistant as it was previously believed. It is reported by many
authors that there is local control of 75%-85%.(19- 21)
In
this study we describe our nine- year experience with the treatment of GCTs of
long bones with different surgical modalities.
Attention was directed towards determination of the rate of initial
local recurrence, factors that might predispose to recurrence and the results
of secondary procedures for the treatment of recurrences.
Methods
Between July 1994 and July
2003, a total of forty-three patients with giant cell tumor (GCT) of the long
bones have been treated at the hospitals of the Royal Medical Services (RMS).
Of these, Thirty-seven were included in the present study
on the basis the documentations available.
All patients were evaluated by
clinical examination, routine laboratory tests, local plain X-ray, chest x-ray,
computerized tomography and magnetic resonance imaging. Biopsy was taken in all
cases to confirm the diagnosis and to define the histological grade of the
tumor. The lesions were classified according to the radiographic parameters
considered by Campanacci et al(7) into grade I, II or
III. Different surgical modalities were used including: curettage with bone grafting; curettage with
bone cement filling; Curettage and Adjuvant with bone cement and / or Bone graft; wide
surgical resection; wide surgical
resection with custom- made total joint arthroplasty; wide surgical resection with arthrodesis.
Curettage was done through a large cortical window by the manual curette and by
the dental burr in some cases. The adjuvant local therapy used in our cases were
hydrogen peroxide (H2O2) and electrical cautery. Selection of the surgical technique was based
on the site and size of the lesion, soft tissue involvement (intra- or
extra-compartmental), tumor grade (histological and radiological) and if
recurrent or not. Patients were followed-up, clinically and radiologically a minimum of two years (2-9
years) to detect local recurrence, pulmonary metastasis, local complications of
surgery and to assess the functional outcomes
the patients.
Results
The mean
age of our patients at presentation was 29.3 years (ranged from 19 to 52Years)
and at last follow up visit 32.1 years (range 24 to 55 years). Seventeen
patients were males and twenty were females, (male to female ratio was (1:1.2).
According to the classification of Campanacci et al,(7)
four patients were grade I, 29 patients were stage II and 4 were grade III) Fig.
1. The distribution of the tumor
according to the anatomical site showed that approximately 62% of the lesions
are located about the knee, at the distal femur
and proximal tibia Table I. Three patients had pathological fractures at the time of
presentation.
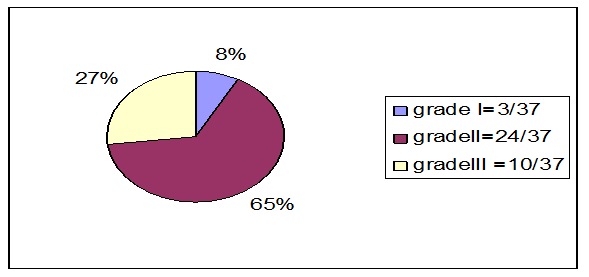
Fig. 1. Distribution of the patients
with GCT according to the radiological grade of Campanacci et al
Table I. Distribution of GCTs
depending on the site
|
Site
of tumor
|
No.
of patients
|
%
|
|
Distal femur
|
12
11
|
23/37
|
32
30
|
62
|
|
Proximal tibia
|
|
Distal radius
|
4
|
10.8
|
|
Proximal humerus
|
2
|
5.4
|
|
Distal tibia
|
2
|
5.4
|
|
Proximal fibula
|
2
|
5.4
|
|
Proximal femur
|
1
|
2.7
|
|
Proximal radius
|
1
|
2.7
|
|
Distal ulna
|
1
|
2.7
|
|
Clavicle
|
1
|
2.7
|
|
Total
|
37
|
100
|
Table II. Distribution of GCT cases
according to primary treatment modality
|
Site of tumor
|
Curettage
+ Bone Graft
|
Curettage
+ Bone Cement
|
Curettage+Adjuvant + bone graft
+ Bone Cement
|
Wide
Excision
|
Wide
Excision +
Reconstruction
|
|
Distal femur
|
4
|
4
|
4
|
-
|
-
|
|
Proximal tibia
|
2
|
4
|
5
|
|
|
|
Distal radius
|
3
|
-
|
1
|
-
|
-
|
|
Proximal humerus
|
-
|
-
|
1
|
-
|
Hemiarthroplasty
1
|
|
Distal tibia
|
1
|
-
|
1
|
-
|
-
|
|
Proximal fibula
|
-
|
-
|
-
|
2
|
-
|
|
Proximal femur
|
-
|
-
|
1
|
-
|
-
|
|
Proximal radius
|
-
|
-
|
-
|
1
|
-
|
|
Distal ulna
|
-
|
-
|
-
|
1
|
-
|
|
Clavicle
|
-
|
-
|
-
|
1
|
-
|
|
Total
|
10
|
8
|
13
|
5
|
1
|
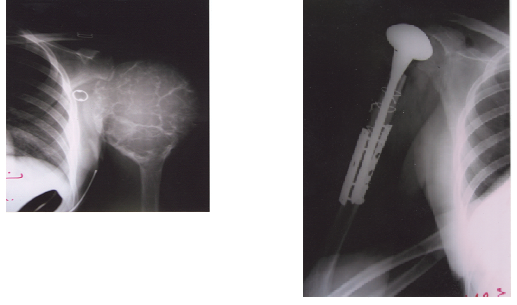
Fig.
2(a,b). GCT
affecting the proximal humerus, large in size, causing severe bone destruction
(a), treated by shoulder hemi- arthroplasty (b)
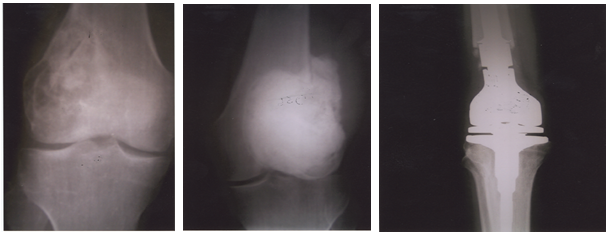
Fig.
3(a,b,c).
Patient with GCT of the distal femur treated primarily by curettage and bone
grafting (a); by curettage and adjuvant therapy with bone cement filling of the
cavity for first recurrence (b); and a third surgery of wide resection and
reconstruction by an endoprosthesis
(custom- made total knee arthroplasty) for the second recurrence (c)
Modalities
of primary treatment we used in our patients are shown in Table II. Six lesions were treated by primary wide
resection; five of them were present at sites where their removal do not
produce significant structural or functional deficit; and one was large in
size, causing sever bone destruction and affecting the proximal humerus. This
female patient was treated by pre-operative embolization, to decrease the
bleeding intra-operatively, and shoulder hemi-arthroplasty Fig. 2. Thirty- one
of our patients were treated by curettage using the manual curette and sometime
the dental burr. Adjuvant therapy (H2O2) was used in
thirteen cases. The cavity was filled with autogenous bone graft, bone cement
or both.
Nine
(24%) of the thirty- seven patients had a local recurrence Table III. All but
one recurrence occurred within three years after the primary operation. The
majority of recurrences occurred in patients with grade III tumors. The three
patients who presented with pathological fracture have developed local
recurrence. Patients who got local recurrence have been treated primarily by
curettage and bone grafting (6 cases) and curettage and bone cement (3 cases).
Lesions managed by curettage and local adjuvant therapy with bone cement and/or
bone graft have no recurrences after a minimum follow-up of two years.
All
patients with local recurrence, except one, have been treated, secondarily, by
curettage (using the burr), adjuvant therapy, bone cement and subchondral bone
graft. One patient, with recurrence in the distal radius, was treated by wide
excision and a wrist arthrodesis (Case No. 7). This patient developed lung
metastasis during follow-up, six months after his second surgery. One patient developed
a second recurrence (Case No. 3), with perforation of the articular surface of
the distal femur, for which a third surgery of wide resection and
reconstruction by an endoprosthesis (custom- made total knee arthroplasty) was
done (Fig. 3). This patient got a fracture of his tibia, at the site of the endoprosthesis,
which was treated conservatively and Primary recurrence in case no. 6 has been treated by wide excision and wrist arthrodesis.
Table III. Data of the patients who had
a local recurrence
|
No.
|
Age,
Sex
|
Site
|
Grade
|
Pathological
Fracture
|
Primary
Treatment
|
Onset
of Recurrence (months)
|
|
1
|
F,
19Y
|
Distal
Femur
|
III
|
Yes
|
Curettage
+ Bone Graft
|
26
|
|
2
|
F,
27Y
|
Proximal
Tibia
|
III
|
No
|
Curettage
+Bone Cement
|
30
|
|
3
|
M,
33Y
|
Distal
Femur
|
III
|
No
|
Curettage
+ Bone Graft
|
12
|
|
4
|
F,
40Y
|
Distal
Radius
|
III
|
No
|
Curettage
+ Bone Graft
|
18
|
|
5
|
M,39Y
|
Distal
Tibia
|
III
|
No
|
Curettage
+ Bone Graft
|
9
|
|
6
|
M,
34Y
|
Distal
Femur
|
III
|
Yes
|
Curettage
+ Bone cement
|
13
|
|
7
|
M,23Y
|
Distal
Radius
|
III
|
No
|
Curettage
+ Bone Graft
|
19
|
|
8
|
F,
24Y
|
Distal
Femur
|
III
|
Yes
|
Curettage
+ Bone Cement
|
15
|
|
9
|
M51Y
|
Proximal
Tibia
|
II
|
No
|
Curettage
+ Bone Graft
|
56
|
Primary recurrences in all
other cases have been treated by Curettage +Adjuvant +Bone graft + Bone Cement Case no. 3 developed a second
recurrence which was treated by wide excision and a custom- made endoprosthesis
(Total Knee Arthroplasty).
healed after 4 months. One patient, with GCT of upper tibia, had
rupture of the patellar tendon 4 weeks post-operatively and reconstruction was
done. None of our patients died during the follow-up period, and amputation was
not done for any.
Discussion
Typically,
GCTs occur in the third and fourth decades of life, and the age distribution in
our series is consistent with that published in previous reports.(7,8)
There were slightly more female patients
(54%) than male patients. This finding is also in agreement with some earlier
studies, but not with others.(6) The tumor is uncommon in
patients with open physis.(7,9) The youngest patient in our
series is 19 year old.
Some
authors(7,14,17,22) have attempted to correlate the clinical
behavior and incidence of recurrence with the initial radiographic grade.
Others (15,18,23) found no correlation between the
radiographic appearance of the lesions and their clinical behavior. Although
statistically insignificant, because of the small number, we observed a strong
correlation in our series, especially for lesions of grade III.
Pathological
fracture might be the primary presentation in 4%-32% of patients with GCTs. Eckardt(22) treated these cases, in his series, by immediate open biopsy
and wide excision. Sung etal(6) and Goldenberg et al,(8)
in two separate studies,have recommended immediate
curettage and bone grafting with additional external fixation. Dreinhofer et
al (10) have treated ten GCTs with fracture at the time
of diagnosis by curettage and bone cementing and reported recurrence in two (20%).
In our study three patients presented with fracture (8%), one has been treated
with curettage and bone grafting and the other two with curettage and bone cementing
and all developed local recurrence.
Because
GCT is a locally aggressive yet benign disease, intralesional treatment is a
limb- sparing option with good outcomes.(1)
Many
different methods of treatment of GCTs
are reported in the current literature including: curettage; curettage and bone
grafting; curettage and insertion of bone cement (polymethylmethacrylate); cryotherapy (liquid nitrogen) after curettage of the cavity; curettage and
a chemical or electrical adjuvant (phenol, zinc oxide, alcohol, H2O2,
argon beam coagulation and electrical
cauterization of the inner surface of the cavity) prior to the insertion of
bone cement or a bone graft; primary resection;radiotherapy; and embolization of the feeding vessels.(6-8,
13-21)
Although
intra-lesional procedures remain the treatment of choice for most GCTs, wide
resection offers the lowest recurrence rate (0-20%) and is recommended for
lesions in certain locations (proximal fibula, proximal radius, distal ulna,
and clavicle) where it leaves no functional deficits.
Table V. The rate of recurrence after
different intralesional treatments of primary GCT of bone (minimum follow-up >
2 years)
|
Author
|
Number
of patients
|
Adjuvant
treatment
|
Rate
of local recurrence
|
|
Goldenberg(8)
|
120
|
None
|
43%
|
|
Campanacci(7)
|
128
|
None
|
30%
|
|
Malawar(13)
|
102
|
Burr, Liquid nitrogen
|
8%
|
|
O’Donell(14)
|
60
|
Burr,
Phenol, Cement
|
25%
|
|
Szendroi(1)
|
11
|
Phenol,
Cement
|
9%
|
|
Blackley(15)
|
59
|
Burr,
None
|
12%
|
|
Mcdonald(18)
|
85
|
Burr,
Phenol, Alcohol
|
34%
|
|
Lausten(24)
|
18
|
None,
Radiotherapy
|
56%
|
Table IV. Recurrence rate in relation
to the primary treatment
|
Primary treatment modality
|
No.
of cases
|
Number
of Recurrence
|
Recurrence
rate
|
|
Curettage + Bone Graft
|
10
|
6
|
60%
|
|
Curettage + Bone Cement
|
8
|
3
|
38%
|
|
Curettage + Adjuvant +
bone graft
+ Bone Cement
|
13
|
-
|
-
|
|
Wide Excision
|
5
|
-
|
-
|
|
Wide Excision +
Reconstruction
|
1
|
-
|
-
|
|
Total
|
37
|
9
|
24% |
However,
in certain sites, wide resection necessitates reconstruction which is
associated with considerable surgical and functional morbidity. In our study,
none of the seven patients who have been treated by wide excision (6 primarily
and one after recurrence) have got recurrence.
Intralesional
curettage and bone grafting is a limb- sparing option that is associated with
good functional outcomes in most cases. However, simple curettage with or
without bone graft has recurrence rate of 27- 55%.(7,8,24) Six of the ten patients we have treated by
curettage and bone grafting developed local recurrence (60%) Table IV.
The high
risk of recurrence led several surgeons to replace bone graft in the lesion
with bone cement packing and to investigate different intralesional adjuvant
therapies. These presumably remove the tumor cells which remain after curettage
because of their thermal or chemical effects.
The
data, from literature, summarized in Table V (as presented in the review
article of Szendroi(1)) suggest that the use of adjuvants
combined with careful curettage may decrease the rate of local recurrence,
which were reported in the historical series of Goldenberg et al (43%)(8) and Campanacci et al (30%),(7)
to about 9% in the series of Szendroi.(1) McDonald et al(18) found in his big series that the
most significant factor in decreasing the rate of recurrence is the surgical
procedure employed for removal of the tumor i.e., curettage with adjuvant
therapy (34%) versus resection (7%). This result has been confirmed by O’Donell.(14) Bone cement technique, compared with bone
grafting, offer the advantages of lack of donor site morbidity, an unlimited
supply, immediate structural stability, low cost, easy to use and contains
barium that sharply contrast the surrounding bone which makes the local
recurrences more readily apparent.
The
disadvantages of using cement include difficulty in removing it in revision
surgery and possibility that subchondral cement may predispose the joint to
early degenerative osteoarthritis. Malawar(13) showed that
subchondral bone grafts are superior to cement for restoration of the normal
subchondral anatomy. We have used curettage and cement in the treatment of
eight patients with 3 recurrences (38%).
Several
authors have added the technique of high speed burring of the cavity after
simple intralesional curettage. A large cortical window is necessary to expose
the entire tumor cavity. O’Donnell(14) and Blackley,(15)
found this technique efficient to decrease the rate of local recurrence to 12%,
17% respectively.
Adjuvant
therapies have advantages and disadvantages. However, they all offer a method
for eradication of microscopic tumor tissue. Recurrence rates with curettage
and phenol 5% and packing with bone cement or bone grafts are 5-17%.(14)
Blackley(15) have raised the concern of the rapid phenol
absorption through cancellous bone and its hazardous effect on the CNS, heart,
kidney and liver.
Many
authors(11,13,17) advocated cryosurgery (liquid nitrogen) as
an adjuvant. They reported 2-12% recurrence rate. Fracture was the most
commonly reported complication. Malawer(13) suggested that
all patients who undergo cryosurgery should receive internal stabilization as
well.
We
did not use phenol, cryosurgery, argon beam, zinc oxide or alcohol in any of
our cases. Curettage followed by high speed burring and H2O2
lavage with bone cement filling with or without subchondral bone graft was used
in 14 patients (13 primary and one recurrent), One of them developed recurrence
(7%).
The
management of local recurrence of GCT varies. Some authors,(7,22,24)
recommend wide excision for any
recurrent lesion, where as others(10,16,17) believe that
rpeated intralesional surgery with adjuvant for the second or third recurrence
is justified. We repeated the intralesional surgery for all except one of our
patients.
Many
authors(19-21) recommend megavoltage radiation as a reasonable
alternative to complex and difficult surgery, especially in areas where surgery
is not accessible or in patients with high risk for surgery .None of our
patients have been treated by this method.
Approximately
three percent of GCTs metastasize to the lung.(3,4) This
complication occurred in one of our cases. One of our patients with a GCT in
the proximal tibia has got a rupture of his patellar tendon which was
reconstructed. Another female patient presented with a subcutaneous soft tissue
nodule with a calcified rim at the scar of the previous surgery.
There
were few patients in each subcategory for us to demonstrate statistical
significance, but the rate of local recurrence seemed to be higher in patients
who had a tumor of the distal radius, those who had an associated pathological
fracture, and those who had a grade- III lesion according to the classification
of Campanacci et al.(7) Use of high speed burr and
adjuvant local therapy may have decreased the rate of local recurrence.(15,23)
Functional outcomes and patients satisfactions were good to excellent, which
were similar to those of many authors.(23, 25-27)
References
1. Szendroi M. Giant cell tumour of bone. J
Bone Joint Surg (Br) 2004; 86: 5-12.
2. Rock MG, Sim
FH, Unni KK, et al. Secondary malignant
giant cell tumor of bone. J Bone Joint Surg(Am) 1986; 68: 1073- 1079.
3. Maloney WJ,
Vaughan LM, Jones HH, et al. Benign metastasizing giant cell tumor of bone.
Clin Orthop 1989; 243: 208-215
4. Bertoni F,
Present D, Sudanese A, et al. Giant cell tumor of bone with pulmonary
metastases. Clin Orthop 1988, 237: 275-285.
5. Kito M, Moriya H, Hikata A, et al. Establishment of a
cell line from a human giant cell tumor of bone. Clin Orthop 1993; 294:
353-360.
6. Sung HW, Kuo
DP, Shu WP, et al. Giant cell tumor of bone: Analysis of two hundred and
eight cases in Chinese patients. J Bone Joint Surg 1982; 64A:755-761.
7. Campanacci M,
Baldini N, Boriani S, et al. Giant cell tumor of bone. J Bone Joint Surg
1987; 69A: 106-114.
8. Goldenberg
RR, Campbell CJ, Bonfiglio M. Giant cell tumor of bone: An analysis of two hundred
and eighteen cases. J Bone Joint Surg 1970; 52A: 619-664.
9. Picci P,
Manfrini M, Zucchi V, et al. Giant cell tumor of bone in skeletally
immature patients. J Bone Joint Surg 1983; 65: 486-490.
10. Dreinhofer
KE, Rydholm A, Bauer HC, et al. Giant cell tumors with fracture at diagnosis. J
Bone Joint Surg(Br) 1995; 77: 189-193.
11. Khalil EA,
Younis A, Aziz SA, et al. Surgical management of giant cell tumor of bones. J
Egyptian Nat Cancer Inst 2004; 18(3): 145-152.
12. Enneking WF. A system of staging
musculoskeletal neoplasms. Clin Orthop 1986; 204: 9-24.
13. Malawar MM,
Bickels J, Meller I, et al. Cryosurgery in the treatment of giant cell
tumor. Clin Orthop 1999; 359: 176-88
14. O’Donell RJ, Springfield DS, Motwani
HK, et al. Recurrence of giant cell tumors of the long bones after curettage and
packing with cement. J Bone Joint Surg
1994; 76(12): 1827-1833.
15. Blackley MB,
Wunder JS, Davis
AM, et al. Treatment of giant cell tumor of
long bones with curettage and bone
grafting. J Bone Joint Surg 1999; 81: 811-820.
16. Yip KM, Leung
PC, Kumta SM. Giant cell tumor of bone. Clin
Orthop 1996; 323: 60-64.
17. Gambini A, Di
Giorgio L, Valeo M, et al. Giant cell tumor of bone. J Orthopaed Traumatol
2003; 3: 126-132.
18. Mcdonald DJ,
Sim FH, Mcloed RA, et al. Giant cell tumor
of bone. J Bone Joint Surg 1986; 68:235-242.
19. Chakravarti
A, Spiro IJ, Hug EB, et al. Megavoltage radiation therapy for axial and
inoperable giant-cell tumor of bone. J Bone Joint Surg 1999; 81(11):
1566-1573.
20. Bell RS,
Harwood AR, Goodman SB, et al. Supervoltage radiotherapy in the treatment of
difficult giant cell tumors of bone. Clin Orthop 1983; 68: 32-36.
21. Kanamori M,
Ohmori K. Curettage and radiotherapy of giant cell tumor. J Orthopaedic
Surgery 2005; 13(2): 171-173.
22. Eckardt JJ,
Grogan TJ. Giant cell tumor of bone. Clin
Orthop 1986; 204: 45- 58.
23. Turcotte RE,
Wunder JS, Isler MH, et al. Giant cell tumor of bone: A Canadian Sarcoma
Group Study. Clin Orthop 2002; 397: 248-258.
24. Laustin GS, Jensin PK, Schiodt
T.
Local recurrences in giant cell tumor of long bones. Int Orthop 1996;
20: 172-176.
25. Davis AM, Bell RS, Badley EM, et al. Evaluating functionl outcome in
patients with lower extremity sarcoma. Clin Orthop 1999; 358: 90-100.
26. Lavoie S,
Turcotte RE, Birthiaume MJ, et al. The arthrogenic effects for
the treatment of (GCT) of the knee. J Bone Joint Surg 1998; 80: 14-16.
27. Trieb K,
Bitzan P, Dominkus M, et al. Giant cell tumor of long bone. J Bone Joint
Surg 2000; 82: 1360-1361.