The first
role in medicine is “first do no harm”. On the surface, adult-to-adult LDLT
disagrees with this principle, because a healthy individual undergoes a major operation
for no direct, physical benefit.(6)
The
primary barriers of Deceased Donor Liver Transplantation (DDLT) in Asian
countries are the cultural and religious beliefs of people for organ donation
after death. Therefore most the patients with liver diseases died while waiting
for liver transplantation.(7)
Still there is considerable
debate concerning donor safety despite great results with LDLT. Risks to the
donor include those associated with invasive pre-surgical testing and the
surgical procedure. These risks are accepted by potential donors when they know
that the patient’s life may be saved without the uncertainty of a cadaveric
waiting list. This study was conducted to
evaluate
the safety and early outcome of donors who underwent partial hepatectomy for
Living donor- related Liver Transplantation at King
Hussein Medical
Center (Amman- Jordan).
Methods
Between
June 2004 to August 2008, a total of 54 candidate donors underwent multistep
evaluation at King Hussein Medical Centre after obtaining approval from Royal
Medical Services Ethical Committee and a written informed consent.
Twenty-six
(48%) were excluded at one step of the evaluation. A total of 28 consecutive LDLTs
(21 right lobes, 5 left lobes and 2 left Lateral lobes) were performed.
Evaluation
and Selection of Donors
The
donor evaluation protocol was designed for testing. It started from simple and
noninvasive to more complex and invasive, assuming continued donor willingness
and lack of contraindications to donation. Testing assured the donor safety and
then evaluated the quality of graft. The minimal age accepted for consideration
was 19 years with the upper age limit 55 years.
The
donor-recipient pair must be blood-group identical or compatible. The donor
evaluation protocol followed at our center is
outlined in Table I.
When
a potential recipient came to our center, he and his family members were
informed of the need for an early liver transplantation, and they agreed to
receive LDLT, and then the risks and benefits of the procedure would be
explained in general. The written informed details focused on the evaluation
protocol, with concentration on invasive testing, surgical procedure, and all
possible risks of the donor hepatectomy. The donor should make the decision voluntarily,
without any emotional pressure.
To
reduce the pressure on potential donors, informed consent was obtained in the
absence of other family members. The donor can withdraw at any time, with the
assurance that an excuse would be provided by the transplant team.
The
evaluation of donors for medical or surgical suitability could be
continued only after
informed consent was made. Acute or
chronic medical illness was excluded by a detailed history and physical examination,
and all donors were screened by laboratory tests including complete blood cell
count, liver and renal biochemistry values, and viral serologic studies.
Positivity of Hepatitis B surface antigen, Human Immunodeficiency Virus antibody,
or hepatitis C virus antibody constituted an outright ineligibility of the
potential donor. Donors with Diabetes Mellitus or hypertension even under
regular control were rejected. The psychological status of the potential donor
was assessed by a clinical psychologist. Abdominal ultrasonography (US) was
performed to evaluate the quality of liver parenchyma, exclude the presence of
tumors, and confirm the patency of blood vessels. Chest radiography and Electrocardiography
were performed to exclude cardiopulmonary disease. Computed tomography (CT), CT
volumetry, multiple detector three-dimensional CT angiography, and three-dimensional
Magnetic Resonance Cholangiography (MRC) were performed to assess liver volume and
identify unsuspected intra-abdominal pathology and anomalous vasculature
incompatible with donation. Liver biopsy was not routinely performed in our
center. If there was radiographic evidence of fatty infiltration or parenchymal
liver disease, even with normal liver function, echo-guided liver biopsy of the
segments to be donated was performed (it was for five donors to rule out
steatohepatitis).
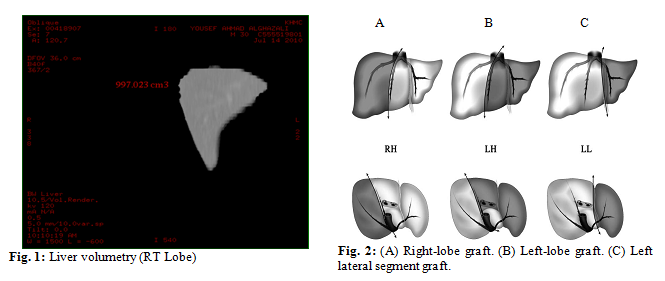
Table III: Underlying diseases of
transplant recipients
|
Recipients
Diseases
|
Number
|
|
Hepatitis B Virus (HBV)
|
7
|
|
HBV+ Hepatocellular carcinoma (HCC)
|
1
|
|
Auto Immune Hepatitis (AIH)
|
6
|
|
Cryptogenic
|
3
|
|
Primary
Sclerosing Cholangitis (PSC)
|
1
|
Progressive Familia Intrahepatic Cholestasis (PFIC)
|
2
|
|
Hepatitis C Virus (HCV)
|
3
|
|
Congenital Hyperbilirubinemia
|
1
|
|
Histocytosis
|
1
|
|
Primary Hyper Oxalosis
|
1
|
|
Biliary Atresia
|
1
|
|
Hepatoblastoma
|
1
|
Table IV: Causes of donor’s exclusion
|
Causes
of exclusion
|
Number
|
|
ABO incompatibility
|
9
|
|
Positive hepatitis serology
|
7
|
|
Liver anatomy anomalies
|
5
|
|
Effects from family, relatives
and society
|
3
|
|
Fatty liver
|
2
|
Table V: Overall donor complications
|
Overall Complications (28.5%)
|
Number
|
Clavien’s classification
|
|
Atelectasis
|
2
|
Grade I:-4(14%)
|
|
Infection
|
2
|
|
|
Pneumonia
|
1
|
Grade II: 2 (7%)
|
|
Bleeding (no re-op)
|
1
|
|
|
Pleural eff. (drain)
|
1
|
Grade III: (7%)
|
|
Biliary leak (drain)
|
1
|
|
|
|
|
Grade IV : 0
Grade V : 0
|
The three hepatectomies are shown in (Fig. 2): right hepatectomy, left
hepatectomy, and left lateral lobectomy.
Follow-up:
Outcomes
related to complications and ongoing symptoms were defined according to Clavien’s classification.(9)
A
specific research assistant was in charge of the whole follow-up. The methods
were taken including record table, telephone follow-up and return visit.
Simple
descriptive statistical methods (frequency, mean and percentage) were used to
describe the study variables
Results
A
total of 54 candidate donors were evaluated for LDLT at our center. Of these, 28
underwent successful hepatectomy for living donation. The mean age of donors
28.89 ± 1.30 (range 19 to 49) years. Twenty-eight donors fulfilled relationship
with the third degree of consanguinity. Demographics characteristics of evaluated
donors are listed in Table II.
The mean age of transplant recipients 35.04 ± 15.58 (range:
3-57 years). The underlying diseases of transplant recipients are listed in Table
III.
A total of 26 potential donors (48%) were
excluded at different points of the work-up. Positive hepatitis serology and
ABO incompatibility were the main contraindications to donation. After the
first step, volunteers withdrew from donation due to effects from family,
relatives and society, with society being some of the reasons for exclusion.
The reasons for exclusion listed in Table IV.
The
mean duration of the operation from skin incision to closure was 6.07± 1.12 (range
4 - 8) hours and the mean intraoperative blood loss was 428.57 ± 297.96 (range 50-
1500) ml. Intraoperative blood transfusion was required for one donor. The mean stay of donors in the intensive care unit
(ICU) was 1.2 ± 0.4 d and the mean hospital stay was 6.43 ± 1.32 (range 5 - 9)
days for left lobe and left lateral donation, and 9.68 ± 2.93 (range 8-20) days
for right lobe donation.
In
the immediate postoperative period, all donors exhibited a significant
transient elevation of liver enzymes and hyperbilirubinemia on postoperative
day one.
Normalization
of serum transaminases and total bilirubin was accomplished by postoperative days
5 to 7. In contrast, prothrombin time exhibited a mild postoperative elevation
that declined to normal level within 3 days.
The
mean follow-up time was 8.54 ± 1.9(range 6- 12) months. Follow-up was not lost
for anyone. The mean recovery time of 28 donors who were followed up for more
than 6 months, was 2.875 ± 0.715 (range 2- 4) months, the mean time to return to
work was 5.0 ± 1.0 months, and 13 of them returned to normal work even earlier.
No
re-operation was performed and no deaths occurred in this series, while morbidity
rate was 8 (28.5%). Four experienced grade I (minor) complications, two
experienced grade II, two experienced grade III, none had grade IV or grade V
according to Clavien classification. Donor
complications are shown in Table V.
Discussion
Liver
transplantation is the only life-saving treatment for patients with end-stage
liver disease. Because of the rarity cadaveric donor organs in Asia, (due to
the cultural and religious beliefs of people with acceptance of brain-death
criteria), and variable shortages in most other parts of the world, the idea of
partial liver donation to help save the life of a family member has
considerable appeal, but reliable information about risks must be provided to
prospective donors. The development of LDLT in Jordan experienced two stages: Pediatric
Living Donor Liver Transplantation (PLDLT) and Adult-to-Adult Living Donor
Liver Transplantation (ALDLT). To ensure the safety of donors, we identified
three basic principles for the selection of donors: independent decision on
donation, no contraindication for donation, and avoidance of obligation in the
process of donation. These principles were strictly fulfilled with no
exceptions.
LDLT
was performed on the premise that the donor liver could be divided into two
separate parts, the remnant liver in the donor would regenerate quickly, and
the donor would not be injured operatively.(10) An essential
part of LDLT is to perform donor hepatectomy with minimum risk while preserving
graft viability.(11) Our operative time was 6.07± 1.12 (range
4-8 hours) and is nearly comparable to the other center 7.6±0.8 hours (range
6.8-10.3 hours).(10)
The
adult-to-child living liver transplantation was first successfully performed in
1989 and accounts for 10% of pediatric liver transplants in the United States,
while ALDLT was first performed in 1993 and accounts for approximately 3% of
adult liver transplants in the United States.(12) Applying the
principle of justice to LDLT is also complex, and nobody knows whether a
procedure that violates the principle ‘above all, do no harm’ can be justified.(13)
Exposing a healthy volunteer to operative insults can be justified only when
donor risk is minimized to an acceptable degree. In practice, complete
prevention of donor complications is not feasible, but many of them appeared to
be prevented or ameliorated.(14)
To
the authors’ knowledge, there have been 12 deaths of right-lobe donors and
three deaths of left-lobe donors worldwide. Additionally, two donors have
required liver transplantation themselves as the result of operative
complications.(15) The overall mortality is 0.2% in relation
to the total number of liver donations worldwide and the risk of death for
donors of a left lateral segment or a left lobe is estimated to be
approximately 0.1%, whereas the risk for donors of a right lobe is estimated to
be approximately 0.4 to 0.5%.(8,16) Donor death has occurred
in both experienced and inexperienced centers. Lack of vigilance and loosening of
acceptance criteria are the major reasons for the donor mortalities. To avoid
further donor death, the transplant surgeon should maintain his role as the
gatekeeper in preventing unjustified and risky donor operations. Finally, he should
have full commitment of life-long and holistic care of the donors.
A
right lobe hepatectomy has a major issue of concern for LDLT to the donors
because of the greater extent of resection and the higher expected risk, and it
is known that the risks associated with right hepatectomy vary and that they
depend primarily on the volume of the remnant left liver, which must be
sufficient so that the donor is not at risk of developing liver failure
post-donation.(5, 17)
Selection
and evaluation of a living liver donor for adult recipients is a complex
process that involves optimizing graft size in relation to the safety of donors
and recipients, technical details of liver procurement, and ethical problems of
using nonrelated live donors, so partial liver donation can be performed safely
with a relatively low-risk of major perioperative morbidity.(1,18)
No
effort should be spared in avoiding complications by appropriate patient
selection, controlling blood loss, meticulous surgical technique, and
postoperative care.(19) However, donor hepatectomy in a
healthy population, should be taken as a situation different from that
encountered in the oncologic field.(20)
In
our center, we strictly followed our protocol; a careful and comprehensive
work-up for selection and evaluation of the donors was made to decrease
mortality and morbidity rate to the range of the other centers in the world.
All our donors returned to their normal life and work.
Conclusion
Donor hepatectomy in living-donor liver
transplantation is a safe procedure. Meticulous and comprehensive selection
protocols are a prerequisite for a good outcome.
References
1.Li F, Yan
L, Zeng Y, Yang J, et al. Donor safety in adult living donor liver
transplantation using the right lobe: Single center experience in China. World
J Gastroenterol 2007; 13(27): 3752-3755.
2.Ng
K, Lo C. Liver transplantation in Asia:
past, present and future. Ann Acad Med Singapore 2009; 38:322-331.
3.Broering DC, Wilms C, Bok P, et al. Evolution of donor morbidity in living related liver transplantation, single-center
analysis of 165 cases. Ann Surg 2004; 240: 1013-1026
4.Soejima Y, Shimada M, Suehiro T, et al. Outcome analysis in adult-to-adult living donor liver transplantation,
using the left lobe. Liver Transplantation 2003; 9(6): 581-586.
5.Cho J, Suh K, Kwon C, et al. Outcome of donors with a remnant liver volume of less than 35% after
right hepatectomy. Liver Transpl 2006; 12:201-206.
6.Robert
S Jr, Brown SR. Live donors in liver transplantation.
Gastroenterology 2008; 134(6): 1802-1813.
7.Sobhonslidsuk
A, Intaraprasong P, Tongprasert S.
Evaluation of donors for living donor liver transplantation (LDLT). J Med
Assoc Thai 2010; 93(5): 637-641
8.Renz
J, Reichert P, Gordon S, Emond J. Surgical
anatomy of the liver. In: R Busuttil and G Klintmalm, editors. Transplantation
of the liver.2nd ed. RD546.T643 2005, and ch 26, Outcomes of living
donor liver transplantation. eg: network Philadelphia: Elsevier, 2005:713-724.
9.Chan
S, Fan S, Lo C, et al. Toward current
standards of donor right hepatectomy for adult-to-adult live donor liver
transplantation through the experience of 200 cases. Ann Surg 2007; 245:
110–117.
10.Wen T,
Chen Z, Yan L, et al. Measures for increasing the safety of donors
in living donor liver transplantation using right lobe grafts. Hepatobiliary
Pancreat Dis Int 2007; 6: 590-595
11.Sugawara Y, Makuuchi M, Takayama T, et al. Safe donor hepatectomy for living related liver
transplantation. Liver Transplantation 2002; 8:58-62.
12.Bramstedt K. Living liver
donor mortality: where do we stand? Am J Gastroenterol 2006; 101:1-5.
13.Nadalin S,
Bockhorn M, Malago´ M, et al. Living donor liver
transplantation. HPB 2006; 8: 10-21.
14.Hwang S, Lee S, Lee Y, et al. Lessons learned from 1.000
living donor liver transplantations in a single center: how to make living
donations safe. Liver Transplantation 2006; 12: 920-927.
15.Florman1
S, Miller C. Live donor liver transplantation. Liver Transpl 2006, 12:
499-510.
16.Walter J, Burdelski M, Bröring C. Chances and risks in living donor liver transplantation. Dtsch
Arztebl Int 2008; 105(6): 101-107.
17.Ibrahim S, Chen C, Wang C, et al. Small remnant liver volume after right lobe living donor hepatectomy.
Surgery 2006; 140: 749-755.
18.Patela S, Orloffa M, Tsoulfasa G, et al. Living-Donor liver transplantation in the United States: identifying donors
at risk for perioperative complications. Am J Transpl 2007; 7: 2344–2349.
19.Azzam A, Uryuhara K, Taka I, et al. Analysis of complications in hepatic right lobe living donors. Ann
Saudi Med 2010; 30(1):18-24.
20.Burcin Taner
C, Dayangac M, Akin B, et al. Donor safety and remnant liver
volume in living donor liver transplantation. Liver Transplantation 2008;
14: 1174-1179.