Abstract
Objectives: To
evaluate, in vitro, the effectiveness of different concentrations of sodium
hypochlorite [NaOCl] (0.5 %, 1.0 %, 2.5% and 5.25% w/v) in the elimination of Enterococcus
faecalis E10e.
Methods: E. faecalis E10e was
grown overnight and allowed to grow until the ‘mid-exponential’ phase. 0.2 ml
of the inoculum that was then transferred into 9.8 ml of NaOCl solution. One ml
samples were removed and poured in 9 ml of sodium thiosulphate [Na2S2O3]
solution (neutralisation) after 30 sec, 1 min, 2 min, 5 min, 10 min and 30 min.
Serial dilution of each neutralised solution was carried out to give a dilution
factor of 10-6. Subsequently, 0.02 ml of each dilution was plated
onto an agar plate, and then incubated at 37°C in an aerobic incubator for 24 hrs. The number
of viable colonies (Colony Forming Units / ml) was determined for each plate.
Results: The results showed that 5.25 % NaOCl was
the most effective irrigant assessed, killing 100% of bacterial cells in 2 min.
However, the time required by 2.5 %, 1.0 % and 0.5% concentrations was 5 min,
10 min and 30 min, respectively.
Conclusion: There was a statistically significant
difference between NaOCl concentrations with respect to the mean number of
viable counts recovered, with 5.25 % NaOCl being the most effective irrigant
assessed. There was a strong curvilinear relationship between NaOCl
concentrations and time taken to attain zero viable counts (100 % killing).
Key words: E. faecalis, Sodium hypochlorite, Viable colonies.
JRMS
March 2012; 19(1): 46-52
Introduction
Sodium Hypochlorite (NaOCl)
has been widely used as an endodontic irrigant since its introduction by Walker
in 1936.(1) When NaOCl
is added to water, hypochlorous acid (HOCl), which contains active chlorine, a
strong oxidizing agent, is formed. Substantial evidence suggests that chlorine
exerts its antibacterial effect by the irreversible oxidation of sulfhydryl
(-SH) groups of essential enzymes, which leads to disturbance of important
metabolic functions of the bacterial cell.(2) Chlorine may
also combine with cytoplasmic components to form N-chloro compounds, which are
toxic complexes and destroy micro-organisms.(3)
The chemical removal of
organic tissue by NaOCl is mediated by the release of hypochloric acid, which
reacts with insoluble proteins to form soluble polypeptides, amino acids and
other by-products.(4) The
clinical efficacy of NaOCl is due to its ability to oxidise, hydrolyse and
osmotically draw fluids out of tissues.(5)
NaOCl is highly effective in
destroying bacteria; however, it does not penetrate well into confined areas of
the root canal system. In addition, it remains in the canals for only a short
period of time, which may limit its effectiveness against bacteria located in
and near the main root canal.(6) Micro-organisms, such as Enterococcus
faecalis, are resistant to NaOCl, especially at low concentrations.(3)
NaOCl is a caustic material
which makes it an effective solvent of both necrotic and vital tissues, but as
it is non-specific agent; it is toxic to the surrounding tissues. Accidental
injection of NaOCl beyond the root apex can cause severe pain, a rapidly
developing oedema, haematoma, necrosis and abscesses.(7)
Oral enterococci,
predominantly Enterococcus faecalis, have been recovered from dental
plaque, saliva, dorsum of tongue, dental caries, osseointegrated dental
implants and persistent root canal infections.(8) The genus Enterococcus
consists of Gram-positive, facultative anaerobic organisms that are ovoid in
shape, non–sporing and may appear on smear in short chains, in pairs or as single
cells.(9) It has the ability to grow in 6.5 % NaCl and pH 9.6,
to grow at 10°C and at 45°C.
Its optimum growth temperature is 35°C. It has a strong reducing ability. It survives at 60°C for 30 minutes and produces ammonia from peptone.(10)
Enterococcus faecalis usually enters the canal during treatment, survives
the antibacterial agents and then persists after obturation.(11)
When E. faecalis is present in low numbers initially, it can be
eliminated. However, once established in
root canal system, it is a difficult organism to eradicate. It can remain
viable, maintains the capability to invade the dentinal tubules, adheres to
collagen in the presence of human serum, acts as a pathogen in failed root
canal treatment.(12) and is one of the few
micro-organisms that have been shown to resist antibacterial effect of calcium
hydroxide..(13,14) The
buffering capability of the hydroxyapetite might protect E. faecalis
from the pH rise by calcium hydroxide.(15) Tissue fluid from the periodontal
ligament and alveolar bone bathing the root of a tooth may provide sufficient
nutrition to E. faecalis within radicular dentinal tubules or the
obturated root canal.(12)
This can enable E. faecalis to grow and gain nourishment
for a long period of time and may account for the presence of E. faecalis
in failed endodontically treated cases.(16) Recognising the potential role of Enterococcus
faecalis in the failure of root canal therapy makes it important to develop
strategies to control infections caused by this organism.
This study was conducted to evaluate,
in vitro, the effectiveness of different concentrations of sodium
hypochlorite [NaOCl] (0.5 %, 1.0 %, 2.5 % and 5.25 % w/v) in the elimination of
Enterococcus faecalis E10e.
Methods
The tested micro-organism
was cultured by placing 50 ml of a fresh nutrient of Fastidious Anaerobe Broth
(FAB; Lab M, Burry, UK) in a sterile 100 ml conical
flask. The broth was then inoculated with Enterococcus faecalis E10e (a
patient isolate that was grown at 37°C for 24 hrs in an aerobic incubator obtained from the
University Dental Hospital of Manchester) using a sterile cotton swab, which
was dipped in the broth before few colonies of the micro-organism were,
harvested from the culture plates. The inoculum was grown overnight in a shaking
water bath at 120 rev/min, 37°C.
A sterile 50 ml conical
flask containing 19.8 ml of (FAB) was inoculated with 0.2 ml of overnight grown
culture (of the same medium) of E.
faecalis E10e and was shaken on a Spinmix (Gallenkamp, Philip Morris
Scientific, Manchester, UK) to ensure thorough mixing.
Aliquots (0.1 ml) of this inoculum was immediately taken and poured into the
first glass bottle containing 0.9 ml of sterile Phosphate Buffered Saline
solution (PBS, Sigma Chemical Co., Steinheim,
Germany) to
give a dilution factor of 10-1. A further five serial dilutions were
made to give a dilution factor of 10-6. This was taken as time zero,
where 0.02 ml of each dilution was removed and plated on Columbia Agar Base
(CBA; Lab M, Burry, UK) plate that had already been
divided into six sections, and then incubated at 37°C for 24 h.
Every hour, 0.1 ml samples
of inoculum from the 50 ml flask (containing the liquid growth medium plus 0.2 ml
of overnight grown culture) in the shaking water bath were taken, serially
diluted, plated as above, and then incubated at 37°C for 24 h. The number of colonies was recorded as
‘viable colony’ with the aid of a plate microscope (Olympus VN, Japan).
The Colony Forming Units per
ml (CFU/ml) were then calculated for each time interval:
CFU/ml = Number of viable
colonies ´ Dilution factor ´ 50.
Exposure to test chemical
(NaOCl):
A sterile 50 ml conical
flask containing 19.8 ml of (FAB) was inoculated with 0.2 ml of overnight grown
culture of the micro-organism tested. The flask was then placed in a shaking
water bath at 120 rev/min, 37°C and the microbial culture allowed growing until the ‘mid-exponential’
phase. At that time, 0.2 ml of the inoculum was removed and poured into a
sterile 20 ml plastic bottle containing 9.8 ml of 0.5 % NaOCl (Sigma-Aldrich
Co. Ltd, Gillingham, UK). One ml samples were removed
and poured in 9 ml of 1.93 g/100 ml sodium thiosulphate solution (Na2S2O3,
5H2O, Sigma Chemical Co., Steinheim, Germany) for neutralisation
after 30 sec, 1 min, 2 min, 5 min, 10 min and 30 min. Immediately, 0.1 ml of
each neutralised solution was taken, serially diluted, plated and then
incubated at 37°C in an aerobic incubator for 24 h. (CFU/ml) were then calculated for
each time interval.
The above experiment was
repeated for other concentrations of NaOCl (1.0 %, 2.5 % and 5.25 %) with the
use of corresponding solutions of Na2S2O3
(1.93 g/100 ml, 1.93 g/100 ml and 3.86 g/100 ml, respectively). The experiment
was repeated four times to achieve statistical significance.
Three controls were included
in this study. The first control was carried out to test if Na2S2O3
could neutralise NaOCl. A sterile 20 ml plastic bottle containing 9.8 ml of 2.5
% NaOCl solution was inoculated with 0.2 ml of fresh sterile medium (FAB). Subsequently, 0.98 ml of this mixture was
added to a sterile 20 ml plastic bottle containing 9 ml of 1.93 g/100 ml Na2S2O3.
Then, 0.02 ml of the ‘mid-exponential’ phase inoculum was added to the bottle,
followed by mixing. Aliquots (0.1 ml) of each neutralised solution was removed
from the 20 ml plastic bottle after 30 sec, 1min, 2min, 5min, 10min and 30 min,
serially diluted, plated and then incubated at 37°C for 24 h. The number of colonies and CFU/ml were
calculated for each time interval.
The second control was
the negative chemical control (no NaOCl solution used) to test if Na2S2O3
by itself was toxic to the micro-organisms used. Aliquots (0.2ml) of the
mid-exponential phase inoculum was removed and poured into a sterile 20 ml
plastic bottle containing 9.8 ml of 1.93 g/100 ml Na2S2O3.
1ml samples were removed and poured in 9 ml of 1.93 g/100 ml Na2S2O3
solution after 30 sec, 1 min, 2 min, 5 min, 10 min and 30 min. Serial dilution
was made to give a dilution factor of 10-6. Subsequently, 0.02 ml of
each dilution was removed and plated on an agar plate and then incubated at
37°C in an aerobic incubator for 24 h. (CFU/ml) were then calculated for each
time interval.
The third control was the
negative microbiological control (no micro-organisms used) to test if the
growth media used were contaminated. Uninoculated broth (0.2 ml) was removed
from a sterile 50 ml conical flask containing 20 ml of (FAB) and poured into a
sterile 20 ml plastic bottle containing 9.8 ml of 2.5 % NaOCl solution. One ml
samples were removed and poured in 1.93 g/100 ml Na2S2O3
(neutralisation) after 30 sec, 1 min, 2 min, 5 min, 10 min and 30 min. aliquots
(0.1 ml) of each solution was taken immediately, serially diluted, plated out
as above and then incubated at 37°C for 24 h. The number of colonies and CFU/ml
was then calculated for each time interval.
Statistical Analysis
The data collected were
entered onto a spreadsheet and statistically analysed using the software
packages (Stata and SPSS/PC + version 10.0).
A regression model was
fitted to the dependent variable viable count, for the independent variables
time and concentration, including an interaction term. In order to take the
clustering of the samples into account, the regression analysis was conducted
using the software package Stata. This package provides robust estimates of the
standard errors of the time coefficient.
The variables time and
viable count were transformed by taking natural logs in base e (and adding one
to avoid taking logs of zero), and this transformation produced a linear
relationship between loge (time + 1) and loge (count +
1), for each concentration. The rest of the analysis was conducted using
SPSS/PC + version 10.0, where descriptive data and plots of the data
were produced.
Results
The number of viable colonies
was recorded and the CFU per ml calculated. The viable counts were the mean of
four repeated experiments performed for E. faecalis E10e. The number of
CFU/ml for E. faecalis E10e at each time interval is shown in Table I.
Exposure to test chemical (NaOCl):
As the concentration of
NaOCl was increased, the time necessary to reduce CFU decreased. At a
concentration of 5.25 %, NaOCl was very effective in killing 100 % of the
tested micro-organism within only 2 min. The CFU/ml for E. faecalis E10e
recovered after exposure to NaOCl is shown in Table II.
Table I. Viable count
determination for Enterococcus faecalis. E10e (CFU/ml)*
|
Time (h)
|
10-4
|
10-5
|
10-6
|
|
0
|
1.015´108
|
5.350´108
|
1.600´109
|
|
1
|
1.075´108
|
6.650´108
|
2.650´109
|
|
2
|
1.120´108
|
7.950´108
|
4.300´109
|
|
3
|
1.335´108
|
9.150´108
|
4.650´109
|
|
4
|
1.415´108
|
1.055´109
|
5.750´109
|
|
5
|
+ +
|
1.170´109
|
7.050´109
|
|
6
|
+ +
|
1.220´109
|
7.650´109
|
|
7
|
+ +
|
1.420´109
|
9.450´109
|
|
8
|
+ +
|
+ +
|
1.140´1010
|
|
9
|
+ +
|
+ +
|
1.305´1010
|
|
10
|
+ +
|
+ +
|
1.370´1010
|
+ +: Number of colonies too many to count
*At 10-1, 10-2, 10-3
dilutions, the number of colonies too many to count
Table II. Viable
count (CFU/ml)* for Enterococcus faecalis E10e recovered after exposure
to (0.5%, 1.0%, 2.5% and 5.25%) NaOCl
|
NaOCl
|
Start (time zero)
|
30s
|
1min
|
2min
|
5min
|
10min
|
30min
|
|
0.5%
|
4.900´109
|
4.050´109
|
3.050´109
|
2.450´109
|
1.600´109
|
6.500´108
|
0
|
|
1.0%
|
8.900´109
|
4.750´109
|
3.150´109
|
2.250´109
|
8.500´108
|
0
|
0
|
|
2.5%
|
9.500´109
|
3.300´109
|
1.950´109
|
3.000´108
|
0
|
0
|
0
|
|
5.25%
|
1.045´1010
|
2.200´109
|
5.000´108
|
0
|
0
|
0
|
0
|
* Average at a dilution factor of 10-6
Table III.
Viable count (CFU/ml) for Enterococcus faecalis E10e recovered after
exposure to
2.5% NaOCl and 1.93g/100ml Na2S2O3 (Control 1)
|
Time
|
CFU/ml*
|
% Survivors
|
|
Start
|
4.700´109
|
100
|
|
30s
|
4.650´109
|
99
|
|
1min
|
4.650´109
|
99
|
|
2min
|
4.650´109
|
99
|
|
5min
|
4.600´109
|
98
|
|
10min
|
4.600´109
|
98
|
|
30min
|
4.500´109
|
96 |
* Average at a dilution factor of 10-6
Control experiments revealed a very high
percentage of survivors even after 30min.
The CFU/ml of the organism tested and the percentage of survivors
recovered after exposures to the mixture (NaOCl + Na2S2O3)
and Na2S2O3
solutions at each time interval were determined (Tables III and IV). All
incubated plates showed no microbial growth following exposure to both NaOCl
and Na2S2O3 solutions, indicating that there
was no contamination of either the growth plates or the liquid growth medium
(FAB).
The results for Enterococcus
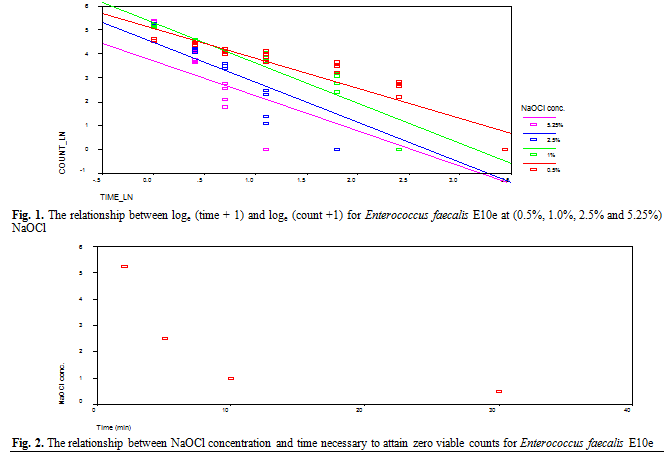
faecalis E10e showed a strong relationship between the mean of viable count
and time, for each concentration of NaOCl solution (Fig. 1). Significant
differences between the concentrations were also apparent for the longer time
intervals (p<0.001), with the highest concentration (5.25 %) reducing the
viable counts to zero in 2 min compared to 30 min for the lowest concentration
(0.5 % NaOCl).
There is, therefore, a
strong curvilinear relationship between concentration and time taken to attain
zero viable counts (Fig. 2).
Discussion
Enterococcus faecalis was chosen for use in this
investigation for various reasons. It is probably the most significant species
in persistent endodontic infections and is the species most frequently found in
cases of apical periodontitis requiring retreatment.(16) It also exhibits resistance
to a wide range of antimicrobial agents, including calcium hydroxide and
Tetracycline.(17,18) and it is easy to grow and identify.(19) Endodontic infections with E.
faecalis usually constitute a problem with treatment because this
micro-organism is difficult to eliminate.(14) Sodium hypochlorite is the
most popular agent for endodontic irrigation, even though its optimum working
concentration has not been universally agreed.(20)
Table IV
Effect of 1.93g/100ml Na2S2O3 on Enterococcus
faecalis E10e (Control 2)
|
Time
|
CFU/ml*
|
% Survivors
|
|
Start
|
8.650´109
|
100
|
|
30s
|
8.600´109
|
99
|
|
1min
|
8.500´109
|
98
|
|
2min
|
8.350´109
|
97
|
|
5min
|
8.350´109
|
97
|
|
10min
|
8.250´109
|
95
|
|
30min
|
8.250´109
|
95
|
*Average at a dilution factor
of 10-6
The
choice of concentration of NaOCl has been a matter of debate, the range
extending traditionally from 0.5% to 5.25%, and a 10% concentration has also
been advocated.(21) The desirable concentration
should be one that possesses low toxicity and adequate antibacterial effects.
Published
work related to the antimicrobial effectiveness of various NaOCl concentrations
has also revealed conflicting results. Some clinical studies have found no
significant difference in antibacterial effect between 0.5% and 5.25% NaOCl.(22) By contrast, another study
has reported that the antibacterial effectiveness of NaOCl is significantly
reduced after dilution.(23)
The
exponential phase of growth is the period of most
rapid reproduction and the one in which the typical characteristics of the
active cell are usually observed.(24) In this study, the “mid-exponential” phase was
selected because microbial population is most uniform in terms of chemical and
physiological properties during this phase.(25)
Viable
count has been developed to count only live cells. The spread plate method was
used in this study for viable count. With this method, a volume of an
appropriately diluted culture is spread over the surface of an agar plate using
a sterile spreader. The plate is then incubated until the colonies appear, and
the number of colonies counted. This method is simple, sensitive and widely
used technique.(26) The method has the
virtue of high
sensitivity; samples containing very few cells
can be counted, thus permitting sensitive detection of microbial contamination
of products or materials.
The
experimental method used in this study was incubation of broth cultures of
selected bacteria with the antimicrobial agent. According to this method, 0.2 ml
of the “mid-exponential” phase inoculum was transferred into bottles containing
9.8ml of 0.5 %, 1.0 %, 2.5 % and 5.25 % w/v NaOCl, respectively. 1ml samples
were then placed in bottles containing 9 ml of neutraliser (sodium
thiosulphate) after time intervals of either 30 s, 1 min, 2 min, 5 min, 10 min
or 30 min to prevent residual action of NaOCl solutions.
Following
neutralisation of NaOCl, 0.1 ml of each neutralised solution was removed,
serially diluted (up to a dilution factor of 10-6) and then 0.02 ml
of each dilution was subcultured in blood agar plate incubated at 37º C for 24
h. The CFU/ml was then calculated.
The results showed that
5.25% NaOCl was the most effective irrigant assessed, killing 100% of bacterial
cells in 2 min. However, the time required by 2.5%, 1.0 % and 0.5%
concentrations was 5 min, 10 min and 30 min, respectively.
The direct exposure method
used in this study appears to be correlated with the irrigant effectiveness and
its direct contact with the micro-organisms. It seems to be independent of
other variables and appears to be a simple, straightforward and practical
laboratory test.(27) However, the clinical efficacy of NaOCl
should be considered in light of the complex root canal anatomy and the
polymicrobial nature of root canal infections.(28) An
irrigating solution that is effective against a single micro-organism tested in
the laboratory may not be effective against a mixed infection in the root canal.(29)
The presence of the smear
layer may prevent the irrigant from penetrating into dentinal tubules in which
micro-organisms may be harboured.(22) Furthermore, interaction with other factors such as organic material, tissue fluids, blood and dentine can influence the antibacterial effectiveness of irrigants.(2) Because chemo-mechanical preparation is a short-time procedure, and NaOCl
remains in the canal for only few minutes, it would appear that the
antibacterial effectiveness of NaOCl inside the root canal might be highly
dependent on its concentration and the time of contact with dentine, organic
matter and pulpal remnants.(6)
Finally, it must be stressed
that the antimicrobial action of NaOCl was tested in this study, but this is
not the only requirement for an endodontic irrigant. Root canal irrigants
should also have other characteristics such as low tissue toxicity, high
detergent power, low surface tension, ease of handling and high proteolytic and
tissue dissolving ability.(30)
The results of controls 1
and 2 showed that high percentage of survivors was recovered even after a
contact period of 30 min. Therefore, sodium thiosulphate solutions were very
effective in the neutralisation of various concentrations of NaOCl and had a
negligible toxic effect on E. faecalis. The results of the third control
showed that there was no microbial growth following exposure to NaOCl and Na2S2O3
solutions. Therefore, no contaminants were found in any of the incubated plates
used in this study.
Conclusions
Under the conditions of this study, it was concluded
that:
·
All concentrations of NaOCl solution were effective in the elimination
of Enterococcus faecalis E10e, but at different time intervals. 5.25 %
NaOCl being the most effective irrigant assessed, producing 100 % killing in 2 min,
followed by 2.5 % (5 min), 1.0 % (10 min) and 0.5 % (30 min), respectively.
·
There was a strong curvilinear relationship between concentration and
time taken to attain zero viable counts (100 % killing).
·
There was a statistically significant difference between the concentrations
with respect to the mean number of viable counts recovered, with 5.25 % NaOCl
being the most effective irrigant used, producing 100 % killing of bacterial
cells in 2 min compared with 0.5 % NaOCl which took 30 min.
References
1. Walker A. Definite and
dependable therapy for pulpless teeth. JADA 1936; 23: 1418-1427.
2. Barber VB, Gomes BPFA, Sena NT, et al. Efficacy
of various concentrations of NaOCl and instrumentation techniques in reducing
Enterococcus faecalis within
root canals and dentinal tubules. International Endodontic Journal 2006; 39: 10-17.
3. Ayhan H,
Sultan N, Cirak M, et al. Antimicrobial
effects of various endodontic irrigants on selected micro-organisms. International Endodontic
Journal
1999; 32: 99-102.
4. Baumgartner
JC, Cuenin PR. Efficacy of several
concentrations of sodium hypochlorite for root canal irrigation. Journal of
Endodontics 1992; 18: 605-612.
5.Huth KC, Quirling M, Maier S, et al. Effectiveness of ozone
against endodontopathogenic microorganisms in a root canal biofilm model. International
Endodontic Journal 2009; 42: 3-13
6.Siqueira
JFJr, Rocas IN, Faviera A, et
al. Chemomechanical
reduction of the bacterial population in the root canal after instrumentation
and irrigation with 1.0%, 2.5% and 5.25% sodium hypochlorite. Journal of
Endodontics 2000; 26: 331-334.
7.Becking AG. Complications in the use of sodium hypochlorite during
enddodontic therapy. Oral Surg Oral Med Oral Pathol 1991; 71: 346-348.
8.Smyth CJ, Halpenny M, Ballagh
SJ. Carriage rates of enterococci in the dental plaque of haemodialysis
patients in Dublin.
British Journal of Oral and Maxillofacial Surgery 1987; 25: 21-33.
9. Murray BE. The life and times of the enterococcus. Clin
Microbiol Rev 1990; 3: 46-65.
10.Sherman JM. The Streptococci. Bacteriol Rev 1937;
1: 3-97.
11.Sjögren U, Figdor D, Spångberg
LS, et al. The antimicrobial effect of calcium hydroxide as a
short-term intracanal dressing. International Endodontic Journal 1991; 24: 119-125.
12.Love R. Enterococcus
faecalis – a mechanism for
its role in endodontic failure. International
Endodontic Journal 2001; 34: 399-405.
13.Safavi KE, Spångberg LSW,
Langeland K. Root canal dentinal tubule disinfection. Journal of Endodontics
1990; 16: 207-210.
14. Kayaoglu G, Erten H, Alcam T, Ørstavik D. Short – term antibacterial
activity of root canal sealers towards Enterococcus
faecalis. International
Endodontic Journal 2005; 38: 483-488.
15.Wang JD, Hume WR. Diffusion of
hydrogen ion and hydroxyl ion from various sources through dentine. International
Endodontic Journal 1988; 21: 17-26.
16. Sedgley CM, Lennan SL, Appelbe OK. Survival of Enterococcus faecalis in root canals ex vivo. International
Endodontic Journal 2005; 38: 735-742.
17. Byström A, Happonent RP,
Sjögren U, et al. Healing of periapical lesions of pulplesss teeth after endodontic
treatment with controlled asepsis. Endod Dent Traumatol 1987; 3: 58-63.
18.Rossi-Fedele G, Roberts AP. A preliminary study investigating the survival
of tetracycline resistant Enterococcus
faecalis after root canal irrigation with high concentrations of
tertacycline. International
Endodontic Journal 2007; 40: 772-777.
19.Ørstavik
D, Haapasalo M. Disinfection
by endodontic irrigants and dressings of experimentally infected dentinal
tubules. Endod Dent Traumatol 1990; 6: 142-149.
20.Sena NT, Gomes BPFA, Vianna ME, et al. In vitro antibacterial activity of sodium
hypochlorite and chlorhexidine against selected single-species biofilms. International
Endodontic Journal 2006; 39: 878-885.
21.Matsumoto T, Nagai T, Ida K, et
al. Factors
affecting successful prognosis of root canal treatment. Journal of
Endodontics 1987; 13: 239-242.
22.Saleh IM, Ruyter IE, Haapasalo
M, et al. Bacterial
penetration along different root canal filling materials in the presence or
absence of smear layer. International Endodontic
Journal 2008; 41: 32-40.
23.Siqueira
JFJr, Batista MD, Frage RC, et
al. Antibacterial effect of
endodontic irrigants on black-pigmented Gram-negative anaerobic and facultative
bacteria. Journal of Endodontics 1998; 24: 414-416.
24.Volk WA, Brown JC. Bacterial
nutrition. In: Basic microbiology. Eighth edition. Addison-Wesley Educational Publishers
Inc., USA.
1997
25.Prescott
LM, Harley JP, Klein DA. Microbial growth. In: Microbiology. Second edition. WC Brown
Communication Inc., USA.
1993
26.Madigan MT, Mortinko JM, Parker J. Brock biology
of micro-organisms. Ninth edition. Chapter 5. Prentice-Hall Inc., New Jersey, USA. 2000
27.Estrela C, Bammann LL, Pimenta
FC, et al. Control of micro-organisms in vitro by
calcium hydroxide pastes. International Endodontic
Journal 2001; 34: 341-345.
28.Spratt
DA, Pratten J, Wilson M, et al. An in vitro evaluation of the antimicrobial
efficacy of irrigants on biofilms of root canal isolates. International Endodontic Journal 2001; 34:
300-307.
29.O’Hara PR, Torabinejad M, Kettering
JD. Antibacterial effects of various
endodontic irrigants on selected anaerobic bacteria. Endod Dent Traumatol
1993; 9: 95-100.
30.D’Arcangelo C, Varvara G,
Defazio P. An evaluation of the action of different root canal irrigants on
facultative aerobic- anaerobic, obligate anaerobic and microaerophilic
bacteria. Journal of Endodontics 1999; 25: 351-353.