ABSTRACT
Objective: To present our experience with elective Endovascular
Aneurysm Repair (EVAR) cases done at King
Hussein Medical
Center, Amman, Jordan.
Methods: This is a retrospective chart review of elective
Endovascular Aneurysm Repair procedures done
during the period of January 2004
to November 2010. One hundred seventeen patients (91 males and 26 females) with
a median age of 71 (range 52 to 79) successfully underwent the procedure. Devices
used included Endurant (Medtronic
AVE, Santa Rosa, Calif), Zenith (Cook Inc, Bloomington,
Ind), Talent (Medtronic), and Excluder (W.L.
Gore, Flagstaff, Ariz). Configurations included, 113 (96.6%) aorto-bi-iliac, three (2.6%) aorto-uni-iliac
with femoro-femoral crossover graft, and one (0.8%) straight tube graft. Most patients
(n=85, 72.6%) scored 3/4 in the American Society of Anesthesia (ASA) scale and
had significant comorbidities. CT
angiography was the main imaging modality used in both preplanning stage and
follow up. Primary outcome measures included overall mortality and freedom from
aneurysm rupture. Secondary outcome measures included access site
complications, graft limb complications, and endoleaks.
Results: No conversions to open
surgical repair or ruptures occurred post graft implantation. A total of 14 (12.0%) patients underwent
re-intervention most of which were for access site problems. These included
wound hematoma treated conservatively (n=5, 4.3%), wound infection treated with
drainage and antibiotics (n=3, 2.6%), and vessel dissection or occlusion
needing surgical repair (n=2, 1.7%). Graft limb complications included
occlusions needing femoro-femoral bypass (n=2, 1.7%). A
total of 27 (23.0%) endoleaks were diagnosed: three (2.6%) were type I and were
managed with ballooning and proximal
extension in one case, 1(0.9%) type 3 managed with an iliac limb extension, and
23 (19.7%) type 2 endoleaks managed conservatively. The mean in hospital length
of stay (LOS) was 3.1 days (range 2-7) while the mean follow up was 16.2 months
(range 6-58). Overall patient mortality during the study period was eight (6.8%) patients
while in hospital mortality was four (3.4%). All late deaths were aneurysm
unrelated.
Conclusion: Endovascular Aneurysm Repair
is technically feasible and effective in aneurysm exclusion and preventing
rupture. It offers major advantages over open aneurysm repair including reduced
early mortality, hospital stay, and use of intensive care facilities. However,
issues of cost effectiveness and late mortality in our setting are still to be
resolved.
Key
words: Aorta, Aneurysm,
Endovascular, Repair.
JRMS December
2012; 19(4): 19-23
Introduction
Repair of aortic abdominal aneurysm (AAA) is performed
to prevent progressive expansion and rupture.(1) The surgical
repair first reported in 1962 remains the treatment with the best long-term results.
It is a major surgical procedure done under general anesthesia, usually
consisting of a mid-line laparotomy and cross-clamping of the aorta and the iliac
vessels. Open surgery has non-negligible mortality (3-7%) and postoperative
complications associated with along hospital stay (10.8 days average).(2)
Since first reported nearly 20 years ago, Endovascular Aneurysm Repair (EVAR)
has been established as a safe and effective alternative to open surgical repair
in the treatment of infra-renal AAAs.(3) Equated to the gold
standard of open repair, EVAR, as a ‘one-time procedure’, substantially reduces
operative morbidity, hospital stay, costs, and utilization of intensive care
facilities if performed in a high-volume center.(4) With
improvements in devices, the main problems with EVAR are being tackled. These
include the need for follow up imaging and re interventions, endoleak, and late
ruptures.
EVAR is best performed in specialized centers. Our
vascular surgery unit is one of the few specialized centers in the region. We
report our experience in EVAR of 117 cases of abdominal aortic aneurysm with
short and medium term outcomes.
Methods
Between January 2004 and November 2010, 117 patients (91
male, 26 female) with a mean age of 71 (range 52 to 79) underwent EVAR at King Hussein
Medical center, Amman, Jordan.
This is a retrospective review of data collected from
patient charts and scans.
Devices used included: Cook Zennith (n=11, 9.4%), Gore
excluder (n=3, 2.6%), Medtronic Talent (n=52, 44.4%), and Medtronic Endurant (n=51,
43.6%). Graft configuration was mainly modular bifurcated aorto-bi-iliac (n=111,
94.9%)) while a straight tube graft was used in one (0.9%) case and
aorto-uni-iliac with femero-femoral cross over bypass was used in five (4.3%)
cases. In five patients (4.3%) there was
hypogastric artery involvement requiring coil embolization.
All procedures were performed by members of the
vascular team including our interventional radiologist in the setting of a
multidisciplinary team. All procedures performed prior to January 2007 were
done in the operating room with C-arm imaging system while procedure planning
involved spiral CT and angiography. All procedures done since were performed in
an interventional suite using a Siemens imaging system while multidetector CT
angiography was the preoperative imaging modality.
Patient demographics, graft, and procedure details
were collected. The American Society of
Anesthesia (ASA) scale was used in grading patient perioperative risk. All
procedures were preplanned and performed on elective basis.
Earlier procedures were done under general anesthesia
with bilateral femoral cut down. Our current practice and most of our
procedures were done with transverse groin incision and when possible
percutaneous contralateral access under regional anesthesia.
Intravenous heparin (80-100 Units per kilogram) bolus
and prophylactic antibiotic were routinely administered early in the procedure.
Graft implantation involves preliminary angiography, marking of renal levels
for proximal landing and deciding on distal landing which was usually in the
common iliac.
Following implantation of the graft components and
ballooning of the fixation sites, angiography was again performed to ensure that
an adequate seal was obtained. A type II endoleak (sac filling from branch
vessels) at the conclusion of procedure was considered acceptable. Once an
adequate technical result was obtained with no type I endoleak (inadequate seal),
the arteriotomies and groin incisions were closed.
Outcome measures included overall survival and freedom
from aneurysm rupture (primary) in addition to early and late complications including
endoleaks (secondary). CT angiography was the main radiological modality used
in the follow up of all cases.
Results are summarized as means and range for
continuous variables while categorical data are summarized as counts or
percentages. Aneurysm related outcomes were reported in accordance with the published standards. Follow-up involved a plain abdominal radiograph (antero-posterior and lateral views) prior to discharge; CT at 6 weeks, 6 months and then yearly; duplex ultrasound at 6 months intervals for sac expansion or substantial endoleak.
Table I. Patient characteristics
|
Characteristic
|
Number (%)
|
|
Male
sex
|
91 (77.8)
|
|
Tobacco
use
|
105 (89.7)
|
|
Hypertension
|
65 (55.6)
|
|
Diabetes
Mellitus
|
23 (19.7)
|
|
Coronary
artery disease
|
68 ( 58.1)
|
|
Peripheral
vascular disease
|
29 (24.8)
|
|
ASA*
2
|
32 (27.4)
|
|
ASA
3/4
|
85 (72.6)
|
*American Society of
Anesthesia
Table
II. Aneurysm characteristics
|
Characteristic
|
Mean (range)/ centimeters
|
|
Aneurysm
diameter
|
6.2 (5.3-9.1)
|
|
Infrarenal
neck length
|
1.7(1.3-2.9)
|
|
Infrarenal
neck diameter
|
2.8 (2.4-3.8)
|
Results
Perioperative patient demographics, co morbidities,
and ASA grade are presented in Table I, while aneurysm characteristics are
presented in Table II.
The procedure was completed in all patients. Patient presentation was variable and
included: asymptomatic aneurysm found incidentally on imaging for another
reason in 84 (71.8%), vague abdominal and/ or back pain in 18 (15.4%),
athero-embolization in 11 (9.4%), other in four (3.4%) patients.
Overall mortality was eight (6.8%) patients with four (3.4%)
deaths within 30 days of the procedure and all prior to discharge from hospital
(n=1 acute mesenteric ischemia, n=1 myocardial infarction, n=1 pulmonary
embolus), and four (3.4%) late mortalities all of which were aneurysm
unrelated.
No conversions to open
aneurysm repair were undertaken
and there were no late aneurysm ruptures. Graft limb occlusion needing fem-fem
bypass graft was done in two (1.7%) patients.
Access site complications included: wound hematoma
treated conservatively in five (4.3%), wound infection treated with drainage
and antibiotics in three (2.6%), vessel dissection and or occlusion needing
surgical repair in two (1.7%). A total of 27 (23.0%) endoleaks were diagnosed: three
(2.6%) type I managed with ballooning in all cases in addition to proximal
graft extension in one case, 1(0.9%) type III managed with an iliac limb
extension, and 23 (19.7%) type II endoleaks managed conservatively (observed
with early CT and later ultrasound). Of
the type II endoleaks 15 (65.2%) had spontaneous resolution while eight (34.8%)
are still being followed up. All patients with type two endoleaks are still
alive and well despite non regression in aneurysm diameter in three (2.6%)
patients.
A re-intervention was undertaken in 14 (12.0%) patients
with most of these related to access site problems.
The mean LOS was 3.1 days (range 2-7). The mean follow up was 16.2 months (range
6-58) with all surviving patients having a minimum of six months postoperative
follow up period.
Discussion
AAA accounts for 1.2% of male and 0.6% of female
mortality, and the third most frequent cause of sudden death after coronary
artery disease and stroke.(2,5,6) The incidence of the disease
in the general population is 60/1000 inhabitants and between 1.8% and 6.6% in
autopsy studies. In studies of natural history of AAA the rate of aneurysm rupture
and death could exceed 60% within three years of the initial diagnosis.(7)
The incidence of abdominal aortic aneurysms (AAA), treated both in
elective and acute setting, has significantly increased over the past decade.(8)
As one of a few specialized vascular surgery units in
the Middle East, we have noticed the marked
increase in diagnosed aneurysms requiring treatment in our center. An aging
population and smoking prevalence are mainly to blame, however the most
important factor has been the prevalent use of diagnostic radiological
modalities especially ultrasonography in health care institutions. This is
reflected in our data by the fact that 72.9% of referrals for abdominal aortic
aneurysm repair are from incidental findings of aneurysms during imaging for
another reason.
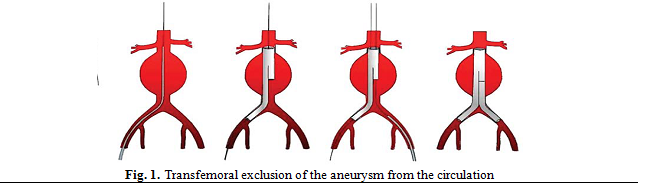
Minimally invasive techniques (EVAR) have been
developed in order to establish an artificial lumen for blood flow that
excludes the aneurysm sac from systemic pressure and such protects from
aneurysm rupture as shown in Fig. 1.(1,2,9)
The obvious advantages that reduce potential
complications of aneurysm repair include absence of laparotomy and aortic cross
clamping. The physiological stress of these two elements on patients is
considerable.
Juan Parodi and colleagues performed the first
endovascular aneurysm repair in Argentina
in1990.(9) Two decades later, the technique has evolved
immensely and new devices are being developed allowing a greater number of
patients to be treated with EVAR.
EVAR is progressively replacing open surgery and now
accounts for more than half AAA repairs in most vascular surgery units.(2,4,10)
Since its introduction, EVAR multiple large clinical trials have
confirmed the perioperative benefits of EVAR compared with open repair
including lower surgical mortality rate.(11)
Although originally introduced for patients considered
unfit for major surgery, EVAR has been used increasingly in patients judged fit
for open repair (OR); results of randomized trials demonstrated that the 30-day
mortality in such patients is around 3%.(9,12,13)
Although we had no open surgical repair controls for
this group of patients, our early mortality rates at 3.4% for aneurysm repairs
are comparable to published data. The late mortality was aneurysm unrelated and
reflects the ASA stage of these patients.
Endoleaks occur when an aneurysmal sac continues to be
pressurized despite endoluminal stent placement. There are a number of types of
leaks: type I (leak at graft ends - inadequate seal), type II (sac filling via
branch vessel e.g. lumbar), type II (leak through a defect in graft fabric),
type IV (a generally porous graft), type V (endotension).(14-16)
Of note is our high rate of endoleaks, especially type
II, comparative to recent published literature. This in part reflects our
learning curve with the different graft types and configurations and in part
due to our early agreed policy of accepting type II endoleaks unless there is
aneurysm expansion on subsequent imaging. Recent literature show that percutaneous
endovascular intervention for type II endoleak even with aneurysm sac growth
does not appear to alter the rate of aneurysm sac growth, and the majority of
patients display persistent/recurrent endoleak.(17) All other
types of endoleak were dealt with ballooning and graft extensions as directed
by the situation.
Patient criteria have been carefully selected
regarding the anatomical suitability for endovascular repair and this is
reflected in our low rate of re-intervention once access site problems were
excluded.
The process of supply of grafts in our programme has
not been optimal. It frequently involved a prolonged application process to an
institutional committee which decides if grafts are bought or not. This also
contributed to our relative long hospital stay where patients are frequently
admitted during the initial application. Another factor contributing to
hospital stay length include receiving patients from remote areas who lack the financial
means for frequent travel to our center and stay as inpatients until the care
plan is finalized.
The required upfront payment of the price of the graft
added to the difficulties of supply and is a relevant issue to most
endovascular practices in developing countries. In comparison, surgical procedures pour into the
local economy with payments distributed between the labor force and local
material supply.
As reported previously, patients undergoing EVAR have
been shown to have a higher quality of life in the short-term when compared
with open repair.(18,19)
Quality of life analysis were beyond the scope of this
publication however all of our patients were discharged home and most report
reasonable early daily activity with no major limitations. Of note also is
patient preference for the procedure once the benefits and risks of both EVAR
and open repair explained.
Conclusions
EVAR is an integral part of aortic aneurysm
management. Reduced early mortality and shorter hospital stay are great
advantages especially in the setting of limited resources and expertise as is
the case in our region. Issues of device supply, cost effectiveness and volume
of practice are subject to further improvement.
References
1.Goncalves FB, Rouwet EV, Metz R, et
al. Device-specific
outcomes after endovascular abdominal aortic aneurysm repair. J Cardiovasc Surg 2010; 51: 515–531.
2.Droc I, Raithel D, Calinescu FB. Endovascular treatment of abdominal aortic aneurysms:
indications and results. Minim Invasive Ther Allied Technol 2011; 20(2):117-124.
3.Brinster CJ, Fairman RM, Woo EY, et
al. Late open conversion
and explantation of abdominal aortic stent grafts. J Vasc Surg 2011; 54:
42-46.
4.Chaikof EL, Brewster DC, Dalman RL, et
al. The care of patients
with an abdominal aortic aneurysm: the Society for Vascular Surgery practice guidelines.
J Vasc Surg 2009; 50(4 Suppl): S2-49.
5.Acosta S, Ogren M, Bengtsson H, et
al. Increasing incidence
of ruptured abdominal aortic aneurysm: a population-based study. J Vasc Surg
2006; 44: 237-243.
6.Alund M, Mani K, Wanhainen A. Selective screening for abdominal aortic aneurysm
among patients referred to the vascular laboratory. Eur J Vasc Endovasc Surg
2008; 35: 669-674.
7.Scheer ML, Pol RA, Haveman JW, et
al. Effectiveness of
treatment for octogenarians with acute abdominal aortic aneurysm. J Vasc
Surg 2011; 53(4):918-925.
8.Cosford PA, Leng GC. Screening for abdominal aortic aneurysm. Cochrane
Database Syst Rev 2007; 18: CD002945
9.Parodi JC, Palmaz JC, Barone HD. Transfemoral intraluminal graft implantation for
abdominal aortic aneurysms. Ann Vasc Surg 1991; 5:496
10.Chaikof EL, Brewster DC, Dalman RL, et
al. SVS practice
guidelines for the care of patients with abdominal aortic aneurysm: Executive summary.
Journal of Vascular Surgery 2009; 50:880-896
11.Jim J, Rubin BG, Geraghty PJ, et
al. Outcome of
endovascular repair of small and large abdominal aortic aneurysms. Ann Vasc
Surg 2011; 25(3):306-14.
12. Prinssen M, Verhoeven EL, Buth J, et
al. A randomized trial
comparing conventional and endovascular repair of abdominal aortic aneurysms. N
Engl J Med 2004; 351:1607–1618.
13. EVAR trial participants. Endovascular aneurysm repair versus open repair in
patients with abdominal aortic aneurysm (EVAR trial 1): randomized controlled
trial. Lancet 2005; 365:2179–2186.
14.Cao P, De Rango P, Verzini F, Parlani
G. Endoleak after endovascular
aortic repair: classification, diagnosis and management following endovascular
thoracic and abdominal aortic repair. J Cardiovasc Surg (Torino)
2010 Feb; 51(1):53-69.
15.Lawrence-Brown MM, Sun Z, Semmens JB,
et al. Type II
endoleaks: when is intervention indicated and what is the index of suspicion for
types I or III? J Endovasc Ther 2009 Feb; 16 Suppl 1:I106-18.
16.Gleason TG. Endoleaks after endovascular aortic stent-grafting:
impact, diagnosis, and management. Semin Thorac Cardiovasc Surg 2009; 21(4):363-372.
17.Aziz A, Menias CO, Sanchez LA, et al. Midterm outcomes of percutaneous endovascular intervention for type II
endoleak with aneurysm expansion. J Vasc Surg 2012; 55: 1263-1267.
18.Hayes PD, Sadat U, Walsh SR, et al. Cost-effectiveness analysis of endovascular versus
open surgical repair of acute abdominal aortic aneurysms based on worldwide
experience. J Endovasc Ther 2010; 17(2):174-182.
19.Madden N, Baril DT, Wertz R, et al. Endovascular abdominal aortic aneurysm repair: a
community hospital's experience. Vasc Endovascular Surg 2009; 43(1):25-29.