Abstract
Objective: To evaluate the effect
of olopatadine 0.1% ophthalmic solution twice daily on ocular and nasal
symptoms in patients with seasonal allergic rhino-conjunctivitis.
Method: This study was
conducted at the Ophthalmology and ear, nose and throat (ENT) clinics on patients
with seasonal allergic rhino-conjunctivitis during spring and early summer seasons
(March to end of June) in 2010 at Princess Haya Al Hussein hospital in the
south of Jordan and in 2011 at Prince Rashid Bin Al Hassan hospital in the
north of Jordan. The patients were divided randomly into 2 groups; group A (50
patients) received Olopatadine 0.1% ophthalmic solution (Patanol) twice daily, group
B (51 patients) received placebo in the form of balanced salt solution. All
patients attended ENT and Ophthalmology clinics weekly for 2 visits; they were
reviewed regarding the improvement of ocular symptoms (itching, redness and lacrimation)
and nasal symptoms (sneezing, itchy nose and runny nose).
Results: In those patients who received Olopatadine 0.1% ophthalmic
solution, after two weeks 98%, 98% and 90% of them showed satisfactory improvement
according to a scale of 1 to 5 marked by the patients for itching, lacrimation
and redness respectively compared to 14%, 12% and 6% in group B respectively
(P-value <0.05). Regarding nasal symptoms 90%, 84% and 78% of patients in
group A showed satisfactory improvement regarding sneezing, running nose and
nasal itching respectively compared to 8%, 16% and 10% in group B (P-value
<0.05).
Conclusion: The treatment of ocular allergy positively impacts
nasal symptoms. The use of ocular solution of Olopatadine 0.1% ophthalmic
solution twice daily has an excellent effect on ocular symptoms and good effect
on nasal symptoms, this effect was more significant at two weeks of
treatment. Olopatadine 0.1% ophthalmic
solution is a well-tolerated drug and may be considered as a primary treatment
for patients with seasonal allergic rhino-conjunctivitis.
Key Words: Allergic rhino-conjunctivitis, Olopatadine.
JRMS June 2013; 20(2): 45-50 / DOI: 10.12816/0000090
Introduction
Allergic rhino-conjunctivitis is a common
disorder in the society that affects different age groups. In USA, it was found
that 15% of population are affected by allergic rhinitis and there is an
increase in prevalence with time,(1) also it is considered a
known cause of seeking medical advice at the primary care clinics.(2)
Although rhino-conjunctivitis may affect any age, yet it is considered a
disease of childhood and adolescence. It
usually shows ocular symptoms like itching, tearing and redness, nasal symptoms
such as nasal itching, running nose and sneezing, and general symptoms such as fatigue
and cough.(1,2) If uncontrolled allergic rhino-conjunctivitis
may have adverse impacts on health- related and economic consequences; it can
lead to learning problems especially in school children, (3) may
worsen the symptoms of asthma,(4) and has major impact on daily
life as it can lead to sleep
disturbances, absence from school
or work and impairment of daily activities like sports.(5) Chronic
allergic rhino-conjunctivitis manifests as either seasonal allergic rhino-conjunctivitis
that accounts for 50-90% of cases or perennial
allergic rhino-conjunctivitis, both seasonal and perennial allergic rhino-conjunctivitis
are IgE-mediated hypersensitivity reaction to specific aeroallergens but vary
according to the duration of exposure.(6,7) The term allergic
rhino-conjunctivitis is preferable to allergic rhinitis because most patients
of allergic rhinitis have ocular symptoms of allergic conjunctivitis.(5)
The mainstay for treatment of patients with allergic conjunctivitis is
prevention by avoidance of exposure to aeroallergens. A lot of topical and
systemic products have also been used for the management of allergic rhino-conjunctivitis.(5,6)
One of these products used to control ocular symptoms of allergic rhino-conjunctivitis
is topical Olopatadine ophthalmic solution. Olopatadine hydrochloride is considered
the first antihistamine used once daily with both selective histamine (H 1) receptor
antagonist and mast cell stabilization effects.(8) It has a
dual action of inhibiting the activity
of the already released histamine on its receptors and suppressing further
release of histamine and inflammatory mediators
from the mast cell.(7) The chemical structure for Olopatadine
is 11 - [(Z) - 3- (dimethylamino) propylidene] - 6- 11 – dihydrodibenz [b,e] oxepin
-2-acetic acid hydrochloride, it is a white water-soluble crystalline powder with
a molecular weight of 373.88.(7,8) There are two
concentrations of Olopatadine ophthalmic preparations: 0.1% used twice daily
and 0.2% used once daily. The aim of this study was to evaluate the effect of
ocular olopatadine 0.1% ophthalmic solution twice daily on ocular and nasal
symptoms in patients with seasonal allergic rhino-conjunctivitis.
Methods
This
study was conducted at the Ophthalmology and ENT clinics on patients with
seasonal allergic rhino-conjunctivitis during spring and early summer seasons
(March to end of June) in 2010 at Princess Haya Al Hussein Hospital in the
south of Jordan and in 2011
at Prince Rashid Bin Al Hassan Hospital in the north of Jordan. All
patients above 14- year of age diagnosed with allergic rhino-conjunctivitis were
recruited. The following patients were excluded: age below 14 years, other
external eye or nasal disease, recent eye surgery, as well as patients who were
receiving anti-allergic therapy during the last month. All patients enrolled in
the study were given numbers starting from 1 according to the time of their
attendance to the clinic, patients with odd numbers were considered as a
control group patient and received balanced salt solution (BSS) eye drop twice
daily, and those with even numbers received olopatadine 0.1% ophthalmic
solution twice daily, the drops trade name was not exposed to the patient. A
total number of 101 patients were enrolled in this study, patients who received
Olopatadine (50 patients) were group A and the remaining 51 patients were group
B. All patients were examined and evaluated by ENT and ophthalmology
specialists to establish the diagnosis of allergic rhino-conjunctivitis before
starting the treatment and the assessment was repeated after one and two weeks
of treatment. During follow up visits (after one and two weeks of treatment)
the improvement of ocular symptoms (itching, redness and lacrimation) and nasal
symptoms (sneezing, itchy nose and runny nose) were evaluated based on a scale marked
by the patient from 0 to 5, where 0 stands for no improvement of the symptom and
5 stand for complete relieve of the symptom. We stressed on the
patient's compliance before and during the follow-up period and patient were
instructed not to take any medications and inform us before using any agents
that may interfere with the results.
Table I: Patients' demography and distribution of
prominent ocular and nasal symptoms among patients
|
group
|
A (50 patients)
|
B (51 patients )
|
Total
(101 patients)
|
|
Mean age(yrs)
|
35.3
|
34.0
|
34.7
|
|
Gender
|
males
|
20
|
22
|
42
|
|
females
|
30
|
29
|
59
|
|
Eye itching
|
(50)100%
|
(51)100%
|
(101)100%
|
|
Lacrimation
|
(46)92%
|
(45)88%
|
(91)90%
|
|
Eye redness
|
(35)70%
|
(37)73%
|
(72)72%
|
|
Sneezing
|
(40)80%
|
(37)73%
|
(77)77%
|
|
Running nose
|
(34)68%
|
(33)65%
|
(67)67%
|
|
Nose itching
|
(31)62%
|
(30)58%
|
(61)60%
|
Table II:
The improvement scale of ocular and nasal symptoms after one weeks of treatment
|
|
Scale 0
|
Scale 1
|
Scale 2
|
Scale 3
|
Scale 4
|
Scale 5
|
|
|
A
|
B
|
A
|
B
|
A
|
B
|
A
|
B
|
A
|
B
|
A
|
B
|
|
Eye itching
|
2%
|
57%
|
6%
|
21%
|
6%
|
8%
|
10%
|
8%
|
12%
|
2%
|
64%
|
4%
|
|
Lacrimation
|
4%
|
45%
|
8%
|
20%
|
10%
|
21%
|
10%
|
10%
|
22%
|
4%
|
46%
|
0%
|
|
Eye redness
|
8%
|
47%
|
10%
|
21%
|
12%
|
20%
|
12%
|
12%
|
16%
|
0%
|
42%
|
0%
|
|
Sneezing
|
6%
|
55%
|
8%
|
23%
|
12%
|
10%
|
12%
|
10%
|
14%
|
2%
|
48%
|
0%
|
|
Running nose
|
8%
|
43%
|
8%
|
18%
|
10%
|
21%
|
10%
|
10%
|
16%
|
8%
|
48%
|
0%
|
|
Nose itching
|
10%
|
57%
|
12%
|
21%
|
16%
|
14%
|
24%
|
4%
|
28%
|
2%
|
20%
|
2%
|
Table III:
The improvement scale of ocular and nasal symptoms after two weeks of treatment
|
|
Scale 0
|
Scale 1
|
Scale 2
|
Scale 3
|
Scale 4
|
Scale 5
|
|
|
A
|
B
|
A
|
B
|
A
|
B
|
A
|
B
|
A
|
B
|
A
|
B
|
|
Eye itching
|
0%
|
55%
|
0%
|
23%
|
2%
|
8%
|
2%
|
8%
|
6%
|
4%
|
90%
|
2%
|
|
Lacrimation
|
0%
|
51%
|
0%
|
23%
|
2%
|
14%
|
4%
|
12%
|
10%
|
0%
|
84%
|
0%
|
|
Eye redness
|
2%
|
51%
|
4%
|
23%
|
4%
|
20%
|
8%
|
6%
|
12%
|
0%
|
70%
|
0%
|
|
Sneezing
|
4%
|
57%
|
2%
|
23%
|
4%
|
12%
|
10%
|
4%
|
10%
|
4%
|
70%
|
0%
|
|
Running nose
|
4%
|
43%
|
6%
|
21%
|
6%
|
20%
|
12%
|
8%
|
16%
|
8%
|
56%
|
0%
|
|
Nose itching
|
4%
|
55%
|
8%
|
23%
|
10%
|
12%
|
12%
|
6%
|
24%
|
4%
|
42%
|
0%
|
Results
The mean age for group A was 35.3 years (range
between15 to 54 years) and for group B was 34 years (range between 14 and 52
years), and the female to male ratio in both groups were 1.5:1 and 1.3:1
respectively; there was no significant statistical difference between the two
groups regarding age and gender nor the distribution of prominent ocular and
nasal symptoms, Table I summarizes patients' demography and the distribution of
the prominent ocular and nasal symptoms among the patients. The most prominent
ocular symptom in our study was eye itching which was seen in all patients,
while sneezing was the most prominent nasal symptom, it was seen in 77% of
patients.
The improvement scale of ocular and nasal
symptoms after one and two weeks of treatment is summarized in Table II and III
respectively. Assuming that scales below three (0, 1, 2) have no satisfactory
improvement and scales 3 or more(3,4,5) have satisfactory
improvement. In group A (Olopatadine group); one week treatment yielded
satisfactory improvement in 86%, 78%, and 70% of patients in regard to eye
itching, lacrimation, and redness respectively compared to 14%, 14%, and 12% in
group B (P-value <0.05). Regarding nasal symptoms 74%, 74%, and 72% of
patients in group A showed satisfactory improvement regarding sneezing, running
nose, and nasal itching respectively compared to 12%, 18%, and 8% in group B
(P-value <0.05). The improvement was more significant after two weeks in
those who received olopatadine 0.1% ophthalmic solution; 98%, 98% and 90% of
them showed satisfactory improvement in eye itching, lacrimation, and redness
respectively compared to 14%, 12%, and 6% in group B respectively (P-value
<0.05). Also 90%, 84%, and 78% of patients in group A showed satisfactory
improvement regarding sneezing, running nose, and nasal itching respectively
compared to 8%, 16%, and 10% in group B (P-value <0.05).
Discussion
The term allergic rhino-conjunctivitis is
preferable to allergic rhinitis because most patients of allergic rhinitis have
ocular symptoms of allergic conjunctivitis,(5) that is
because of the strong anatomical and physiological relationship between the
conjunctival and the nasal mucosa through the connection made by the naso-lacrimal
duct.(9,10) These structures share blood vessel network,
lymphatic tissue and system, and nerve networks,(11,12) this explains
the usual co-existence of the allergic process in both the conjunctiva and
nose. For example Pelikan reported the causal role of nasal allergy in cases of
allergic conjunctivitis,(9) as well as allergic kerato-conjunctivitis.(13)
Gomes et al,(14) concluded that there is a central role of the eye in
initiating and propagating the allergic reaction to involve the nose. There was
no significant statistical difference between the two groups regarding age and gender
nor the distribution of prominent ocular and nasal symptoms. Although allergic rhino-conjunctivitis
is more prevalent in children (40%) than adults (10-30%),(6) we
decided to include only patients above 14 years of age as younger children were
found much less cooperative in understanding the grading scale utilized in this
study. Patients with any nasal pathology were also excluded because they may
have symptoms that mimic those symptoms of allergic rhino-conjunctivitis.
It was clear that ocular symptoms were
more prevalent than nasal symptoms in patients with allergic rhino-conjunctivitis,
probably because ocular surface is more exposed to environmental allergens. This
finding was shared by other investigators.(9,13,14) Assuming
that scales below three (0, 1, 2) have no satisfactory improvement and scales 3
or more (3,4,5) have satisfactory improvement. In group A (Olopatadine
group); one week follow-up yielded satisfactory improvement in 86% of patients
with complete relieve of eye itching in 64% of patients. In addition to that 78%
and 70 % of patients had satisfactory improvement regarding lacrimation and eye
redness respectively. The improvement was more significant after two weeks and improvement
in eye itching, lacrimation, and redness was seen in 98%, 98%, and 90% of
patients respectively. This proves that Olopatadine 0.1% ophthalmic solution has
more significant good response after two weeks use. Other studies also focused on
the effect of Olopatadine on ocular symptoms and found that Olopatadine was clinically
superior to the other anti-allergic agents because of its strong dual action of
antihistaminic and mast cell stabilizing properties.(15) Regarding
nasal symptoms, our study showed that Olopatadine 0.1% ophthalmic solution has also
an effect on nasal symptoms; after one week 74% of patients showed satisfactory
improvement in sneezing and running nose and 72% of patients showed
satisfactory improvement in nasal itching. While after two weeks 90%, 84%, and
78% of patients showed satisfactory improvement in sneezing, running nose, and
nasal itching respectively. This also support the idea that hypersensitivity
reactions of allergic rhino-conjunctivitis
start primarily in the conjunctival mucosa that resulting in activation of inflammatory cells mainly mast cells and
their migration through the blood stream into the nasal mucosa making benefit
of the vicinity and sharing of blood vessels network, in addition to
inflammatory mediators released from conjunctival mast cells pass with tears (16)
via the naso-lacrimal duct and contribute to the nasal mucosa
reaction. Activation of mast cells in conjunctiva and nasal mucosa will cause
the release of inflammatory mediators like cytokines, chemokines and other chemotactic
factors that will be responsible for the allergic reaction. The improvement of
the nasal symptoms after installation of olopatadine 0.1% ophthalmic solution
can be explained by two mechanisms; first is inhibition of conjunctival mast
cells that is thought to be the initiating factor of the allergic reaction and second
is due to ante grade migration of olopatadine 0.1% Ophthalmic solution through
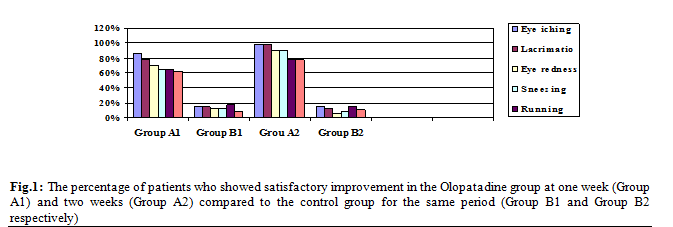
the naso-lacrimal duct to the nasal mucosa. Figure 1, graphically shows the
satisfactory improvement in the Olopatadine group compared to the control group
at one and two weeks. The above results showed that the use of ocular solution
of Olopatadine 0.1% twice daily has an excellent effect on ocular symptoms and
good effect on nasal symptoms. This study supported the results obtained by
other studies in which they studied the efficacy of Olopatadine on ocular and
nasal symptoms, for example Abelson et al,(17)
studied the efficacy
and safety of ophthalmic solution of olopatadine on nasal symptoms in
subjects with history of seasonal allergic conjunctivitis or rhino-conjunctivitis
in spring and fall seasons, he showed that Olopatadine significantly reduced
the frequency of pollen effects on nasal symptoms. Some studies compared the
effect of the two concentrations of Olopatadine and showed that there was no
significant difference in their effect.(18) It is worth to
mention that only 2 patients (4%) in group A and none in group B showed eye
discomfort and dryness, this emphasizes that Olopatadine is well tolerated drug,
this was also concluded by Mah et al.(19) The
only limitation in this study was its dependence on subjective judgments of the
patients which may vary from person to
another, however this study showed clearly the effectiveness of Olopatadine ophthalmic
solution on ocular and nasal symptoms in patients with allergic rhino-conjunctivitis.
Conclusion
The treatment of ocular allergy
positively impacts nasal symptoms. The use of ocular solution of Olopatadine
0.1% ophthalmic solution twice daily has an excellent effect on ocular symptoms
and good effect on nasal symptoms, this effect was more significant at two
weeks of treatment. Olopatadine is a well
tolerated drug and may be considered as a primary treatment for patients with
seasonal allergic rhino-conjunctivitis.
References
1. Meltzer EO.
The prevalence and medical and economic impact of allergic rhinitis in the United States.
J Allergy Clin Immunol. 1997; 99(6 pt 2):S807.
2.Gregory C, Cifaldi M, Tanner LA.
Targeted intervention programs: creating a customized practice model to improve
the treatment of allergic rhinitis in a managed care population. Am J Manag
Care. 1999; 5:485- 496.
3. Baatenburg de JA, Rijdt
JP, Brand PLP. Cough and malaise in young children
due to allergic rhino-conjunctivitis. Ned Tijdschr Geneeskd. 2005;
149:1545- 1548.
4.Kocevar SV, Thomas J,
Jonsson L, et al. Association between
allergic rhinitis and hospital resource use among asthmatic children in Norway. Allergy.
2005; 60:338- 342.
5.Hans DG, Paul LB, Wytske
FF, et al. Allergic rhino-conjunctivitis in
children. BMJ 2007; 335(7627): 985–988.
6.Wallace DV, Dykewicz MS. The diagnosis and
management of rhinitis: An updated practice parameter. J Allergy Clin
Immunol. 2008; 122: S1-84.
7. Bielory L. Allergic diseases of the eye. Med Clin
North Am. 2006; 90: 129-48.
8.Abelson
MB,
Gomes PJ. Olopatadine 0.2% ophthalmic solution: the first
ophthalmic anti- allergy agent with once-daily dosing. Expert Opin Drug Metab
Toxicol. 2008; 4(4): 453- 461.
9.Pelikan Z.
The causal role of the nasal allergy in some patients with allergic conjunctivitis.
Allergy 2002; 57 (Suppl 73):230.
10.Leonard B. Allergic and
immunologic disorders of the eye. Part II: Ocular allergy. J Allergy Clin Immunol.
2000; 1019- 1032.
11.O'Sullivan NL, Montgomery
PC, Sullivan DA. Ocular mucosal immunity. In: Mestecky
J, Binnenstock J, Lamm M, et al (eds). Mucosal immunology, 3rd ed.
Elsevier-Academic Press: Burlington (MA, USA), San Diego
(CA, USA), London, 2005; 1477–1496.
12. Motterle L, Diebold Y, De Salamanca AE,
et al. Altered expression of neurotransmitter receptors and
neuromediators in vernal keratoconjunctivitis. Arch Ophthalmol 2006;
124: 462–468.
13.Pelikan
Z .The possible involvement of nasal allergy in allergic
keratoconjunctivitis. Eye. 2009; 23: 1653–1660
14.Gomes PJ, Krokhmal L, Abelson MB,
et al. Allergic rhino-conjunctivitis is mediated primarily
through ocular exposure to allergen. Invest
Ophthalmol Vis Sci
2004; 45.
15. Lanny J, Terrence O, Jonathan W.
Mast cell stabilization and antihistamine effects of olopatadine ophthalmic
solution: a review of pre-clinical and clinical research. Current Medical Research
and Opinion 2005; 21(9): 1377–1387.
16. Pelikan Z. Conjunctivitis – A Complex
and Multifaceted Disorder. Croatia: In Tech
publications; 2011; p57-74. Available on line: http:
//ebookee.org/Conjunctivitis-A-Complex-and-Multifaceted-Disorder_1554047.html
17.Abelson MB, Gomes PJ, Vogelson CT,
et al. Effects of a new formulation of olopatadine ophthalmic
solution on nasal symptoms relative to placebo in two studies involving
subjects with allergic conjunctivitis or rhino-conjunctivitis. Current Medical
Research and Opinion 2005; 21(5): 683–691.
18. Abelson
MB.
Efficacy of once-daily olopatadine 0.2% ophthalmic solution compared to
twice-daily olopatadine 0.1% ophthalmic solution for the treatment of ocular
itching induced by conjunctival allergen challenge. Current Eye Research
2007; 32:1017–1022.
19. Mah FS, O’Brien T, Kim T,
et al. Evaluation of the
effects of olopatadine ophthalmic solution, 0.2% on the ocular surface of
patients with allergic conjunctivitis and dry eye. Current Medical Research
and Opinion 2008; 24(2): 441–447.