Introduction
The term
‘temporomandibular disorders' (TMD) is used to describe a number of clinical
problems that
involve the temporomandibular joints (TMJ) or masticatory muscles or
combinations of both.(1) Most patients with this diagnosis suffer from
muscle and/or joint pain on palpation and/or mandibular movements.
Additionally, joint sounds may occur and the mandibular range of motion may be
limited.
The
prevalence of TMD has been extensively reported in the literature and several
indices and criteria have been developed.(2) The literature reports great variability in
the prevalence of clinical symptoms (6-93%) and signs (0-93%), probably as a
result of the different clinical criteria used.(3) A simple
comparison is difficult because of the lack of uniform criteria. One of the
most widely used indices is that developed by Helkimo (1974) which combined
anamnestic and clinical dysfunction indices.(4)
Many studies
reported prevalence with different age groups in many countries. Thilander et
al. reported that one or more
clinical signs were reported in 25% in 5-17 year olds,(5)
while more recent studies carried out in Saudi Arabia reported the prevalence
of TMJ signs around 20% and symptoms 24-33%
in 12-16 year old children.(6,7) A high prevalence of 68% was reported in a
Brazilian study among university students,(8) while a Japanese
study, reported a 74% prevalence in the same age group.(9)
One geriatric study reported that objective signs and symptoms are more often
reported than younger subjects.(10)
The aetiology of TMD remains a subject of controversy
and is generally viewed as multifactorial. Nevertheless, a number of studies
have implicated occlusal interferences and psychological factors as more
important than other variables in providing explanation for TMD.(11-14)
Choi et al. reported that the Prior experience of a dislocated disc was
found to be the most risky factor in TMD. Stress was related to limitations of mouth
opening, and the experience of trauma in the TMJ was found to be related to
pain in the joint region. Subjects with high sleep bruxism activity tend to
feel more stressed at work and in their daily life, which in turn might
influence their physical state. These subjects also seem to deal with stress in
a negative way.(15) Bruxism may not be a direct risk factor
in TMD, and the clenching habit found to
be more harmful
than bruxism.(16) The relationship between bruxism and
temporomandibular disorders, if it exists, seems to be
controversial and unclear.(17) Pergamalian et al.
reported that tooth wear factor did not differentiate patients with bruxism
from those without and the amount of bruxism activity was not associated with
more severe muscle pain and was associated with less pain in the TMJ on
palpation.(18) Other investigators have looked at the
correlation between TMD and orthodontic treatment. However, these correlations
have not been clearly established.(19- 22)
While the
literature abounds TMD in developed and some developing countries, very little
has been reported in Arab countries. To the best of our knowledge, there are no
such reports in Jordan.
The purpose of this study is therefore to determine the prevalence of signs and
symptoms of TMD among young adults Jordanians.
Methods
A group of young adult Jordanians were presented to Officers
Election Committee for Mu’tah Military University, from different provinces of Jordan. All the
subjects had just finished their high school examination which is considered a
university entrance examination. Their ages were 18± 6 months.
A questionnaire was designed to assess the anamnestic
and clinical dysfunction indices according to Helkimo.(4) The anamnestic
examination was based on the reported symptoms by the subjects and classified
according to the anamnestic dysfunction index (Ai) as 0, I, or II. While Ai0 comprised
individuals with subjectively symptom-free TMD, AiI and AiII represent those
presented with mild and severe symptoms, respectively.
The clinical examination was based on maximum
mandibular opening, and maximum eccentric mandibular movements during protrusive
and lateral movements, these movements were measured in millimetres. Those
measurements were obtained by using a digital calliper with a sensitivity of
0.01mm. Each movement was
repeated three times in order to obtain an average of the values.
The temporomandibular joint was examined for sounds
and pain. Auscultation of
articular sounds was carried out with
the aid of a stethoscope positioned on
the TMJ lateral region, while the volunteer was performing mouth opening and
closing movements, consecutively and uninterruptedly three times, in order to
observe the presence of articular sounds. TMJ pain was assessed by palpating
the TMJ on rest and during movement and was reported as present or not.
The
muscles of mastication (masseter, temporalis, and medial pterygoid) were
palpated for tenderness. In addition, the lateral pterygoid was examined
against forced contraction. Depending on the clinical dysfunction score (CDS)
following clinical examination, each subject was classified as having a
clinical dysfunction index (Di0) of (0 points) for individuals with clinically
symptom-free TMD, DiI (1-4 points) for those
with mild symptoms, Di II (5-9 points), and Di III (10-25
points) for individuals with moderate and severe TMD symptoms, respectively. No
reference was made to the occlusal component in this study.
All the examinations were performed by one examiner
who was trained and calibrated in the use of the index. The use of one examiner
would insure the continuity of interpretation of the answers provided by the
subjects. To confirm intra-examiner reliability 48 subjects were randomly
selected and re-examined in the same day to reduce the risk of symptom
fluctuation.(23) Dahlberg's
formula was used to calculate the
standard error of the method, and Houston
coefficient of reliability(24) was calculated. The maximum mandibular opening error was 0.34mm, for the
maximum right lateral movement was 0.39mm, for the
maximum left
lateral movement was 0.37mm, and for the maximum
protrusive movement was 0.31mm. The Houston's
coefficient of reliability was above 92% for all the above measured variables.
Data were collected, tabulated, and statistical
analyses were performed using Statistical
Package for Social Sciences (SPSS for Windows, SPSS, Chicago, III). Chi-square
test was used to compare sex differences in both anamnestic and clinical
dysfunction index scores. Correlation coefficients between both scores were
calculated.
Results
A total of 5,426 Jordanians 3,916 males (77.9%), and 1,510
females were presented to Officers Election Committee for Mu’uta Military
University, from different provinces of Jordan. (27.1%) of them 114
subjects (69 females, 45 males) having history of orthodontic treatment were
excluded from this study. The remaining 5,312 subjects 3,871 (72.9%) males, 1,441
(27%) females were included.
The data were pooled in the present investigation. As there were no
statistically significant differences between genders regarding reported
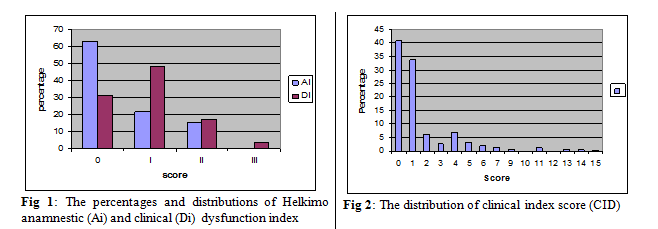
symptoms (P=0.31) or clinical signs (P=0.27). Whilst about 63% (n=3274)
reported no symptoms (Ai 0), 1118 subjects (21.5%) reported mild symptoms (Ai
I) and 806 subjects (15.5%) had severe symptoms (Ai II). Similarly, 1,622 subjects (31.2%) showed no
signs of dysfunction (Di 0);
2,489 subjects (47.9%) had mild signs (Di I); 889
subjects (17.1%) showed moderate signs (Di II); and 198 subjects (3.8%) showed
severe signs of dysfunction (Di III; Fig. 1).
The
majority (91.2%) of the subjects’ demonstrated maximal opening capacity (40mm
or more) while 8.8% demonstrated restricted vertical movement. Similarly, about
one fourth (24.6% right; 26.1% left) demonstrated restricted lateral mandibular
movement, while 37.7% showed restricted protrusive mandibular movement as shown
in Table I.
An analysis
of the signs of the TMD showed that joint sounds was the most frequently
recorded sign (48.2%) among Jordanians followed by impaired range of movement
(15.4) as shown in Table II.
Furthermore, the clinical dysfunction scores showed that 59.2% of the sample
presented with one or more clinical signs of dysfunction. The highest recorded
score was 15 points (Fig. 2). The linear correlation coefficient (r) between
the reported symptoms (Ai) and the recorded
signs (Di) was
0.32, and between the reported
symptoms (Ai) and
the
clinical dysfunction score (CDS)
was 0.37. Although these values were low,
they were statistically
significant (p=0.021).
Discussion
The
population group was chosen for this study for two reasons. First,
almost all of them were borne in the same year (1987) having the same age (18
years±6 months) making it nearly an ideal sample regarding the sample size
and the age group. Second, they belong to different provinces in Jordan
making the sample very close to be representative to Jordanian population at
this specific age group.
The number of 18 years old Jordanian population were
about 98.000 in the year when this study was conducted, they were distributed
almost equally between both genders.(25) The sample size
included in this study represented about 5.5% of the population of that
particular age group.
The lack of differences in the reported symptoms and
clinical signs as revealed in this study tends to agree with other
investigators.(26,27)
Although other studies found a higher incidence of TMD in females.(28-30) This may be
attributed to the different criteria and different age group than those used in
this study.(28,31)
Perceived symptoms of the TMD (anamnestic index) in
the present study conform with the generally reported values(31,32)
despite the 57% and 12% values reported by Helkimo (1979)(4) and Abdel-Hakim
(1983),(33)
respectively. The fact that joint sounds were the commonest specific symptom in
this study was in agreement with other studies reported in the literature,(14,34) although other
criteria were used to evaluate joint sounds than those used in this study.
Regarding the prevalence of clinical signs of TMD,
wide variations were reported in literature. While Mazengo and Kirveskari reported
40% and 37%, respectively,(35) Helkimo and Carlson reported higher
values of 61% and 73%, respectively.(4,36) The
relatively high value reported in this study (59.2%) may be largely attributed
to the fact that about half (48.2%) of the subjects appeared to have joint
sounds. In addition, about one forth of the sample demonstrated restricted
lateral movement and more than one third (37.7%) demonstrated restricted
protrusive mandibular movement. Although there is no obvious reason to explain
these results, but they should be interpreted with caution simply because
considerable proportion of the subjects are not accustomed to making such
movement.
Pain is known as an important feature of TMD because
it is the most important reason for seeking treatment for TMD Greene et al.(37) Between 3-7% of
the population seeks treatment for pain and dysfunction.(3)
Isong et al. reported the TMD type of pain
differ significantly by race, age, and gender after adjusting for socioeconomic
status.(38)
In contrast to other studies, the results of this
investigation revealed that only about 13% of the subjects suffered from pain. This
may have been due to subjects hiding their actual complaint of pain because
they might think it may negatively impact the committee's decision to accept
them as officers. Hence, may not reflect the actual distribution of pain among
this particular sample.
Low correlation coefficients between reported symptoms
and recorded signs as well as the clinical dysfunction score do not necessarily
indicate that significant proportion of the sample have TMD. As these results
can be interpreted in a different way.
For example the considerable proportion of the sample demonstrated
restricted protrusive and lateral movements may not reflect an actual problem
as there are no studies known to determine the normal range of lateral and
protrusive movement in Jordanian population.
Although the Helkimo index has been widely used, it
suffers from some limitations. Most importantly is the issue of validity,
making its general applicability difficult. It would appear that a rather
arbitrary cut-off points and values for different classes within the subscale have
been selected, yet not weighted accordingly. In addition, the issue of
unidimensionality within the index should be resolved. Van Der Weele and Dibbet(39) stated that even
though the index may be acceptable as a valuable instrument in assessing the
TMD, much is still needed to improve the existing scale.
Conclusions
There were no statistically significant differences
between both sexes regarding reported symptoms or clinical signs.
In general the prevalence of clinical signs and
reported symptoms were comparable to other West European studies. TMJ sounds
was the most prevalent sign in contrast to the West European studies that
showed pain as the most frequently reported sign.
References
1.Okeson J. Orofacial pain. IL, USA:
Quintessence Publishing; 1996.
2.Nilner M, Lasing D. Prevalence of
functional disturbances and diseases of the stomatognathic system in 7-14 years
old. Swedish Dental Journal 1994; 5: 173-187.
3.Roda PR,
Fernandez D JM, Bazan HS, Soriano JY. Review of tempromandibular joint pathology. Part 1: classification, epidemiology and risk
factors. Med Oral Patol Oral Cir Bucal 2007; 12 (4): E292-8.
4.Helkimo M. Tembromandibular
joint. Function and dysfunction (eds G.A. Zarb and G. E. Carlson, Munksgaard, Copenhagen, 1974: 175.
5.Thilander
B, Rubio G, Pena L, De Mayorga C. Prevalence of
tempromandibular dysfunction and its association with malocclusion in children
and adolescents: an epidemiologic study related to specified stages of dental
development. Angle Orthodontist 2002; 27 (2): 146-154.
6.Rabab MF. Signs and symptoms of
tempromandibular disorders and oral parafunctions in urban Saudi Arabia adolescents: a
research report. Head and Face Medicine 2006; 25 (2): 1-7.
7.Faris NMA. Symptoms and signs of
tempromandibular disorders and oral parafunctions among Saudi children. J Oral Rehab 2003; 30:
1200-1208.
8.Pedroni
CR, De Oliveira AS, Guartini MI. Prevalence study of signs and
symptoms of tembromandibular disorders in university students. J Oral Rehab
2003; 30: 283-289.
9.Miyake R,
Ohkubo R, Takehara J, Morita M. Oral parafunction with symptoms of tembromandibular
disorders in Japanese university students. J Oral Rehab 2004; 31: 581-523.
10.Schmitter M,
Pammerlsberg P, Hassel A. The prevalence of sign and symptoms of
tempromandibular disorder in very old subjects. J Oral Rehab 2005; 32: 467-473.
11.Mohlin B, Pilley RJ, Shaw WC. A survey of
craniomandibular disorders in 1000 12- years-olds. Study design and baseline
data in a follow up study. European Journal of Orthodontics 1991; 13:
111-123.
12.Egermark I, Thilander B. Craniomandibular
disorders with special references to orthodontic treatment: an evaluation from
childhood to adulthood. American Journal of Orthodontics and Dentofacial
Orthopedics 1992; 101: 28-34.
13.Mintz SS. Craniomandibular
dysfunctions in children and adolescents: a review. Journal of
Craniomandibular Practice 1993; 11: 224-231.
14.Verdonk A, Takada K, Kitai N, et al. The prevalence of
cardinal TMJ dysfunction symptoms and its relationship to Occlusal factors in
Japanese female adolescents. J Oral Rehab 1994; 21: 287-297.
15.Giraki M, Schneider C, Schäfer R, et al. Correlation between
stress, stress-coping and current sleeps bruxism. Head Face Med 2010 Mar; 5: 6: 2.
16.Chio YS,
Chouang PH, Moon HS, Kim SJ. Tempromandibular disorders in 19-years-old Korean
men. J Oral Maxillofac Surg 2002 July; 60(7): 797-803.
17. Barbosa Yde
S, Miyakoda LS, Pocztaruk Rde L, et al. Temporomandibular disorders and bruxism in
childhood and adolescence: review of the literature. Int J Pediatr Otorhinolaryngol 2008 Mar; 72(3):299-314. Epub 2008 Jan 3.
18. Pergamalian
A, Rudy TE, Zaki HS, Greco CM. The association between wear facets, bruxism,
and severity of facial pain in patients with tempromandibular disorders. J
Prosthet Dent 2003Aug; 90 (2): 194-200.
19.Reynder RM. Orthodontics and
tembromandibulars: a review of the literature (1966-1988). American Journal
of Orthodontics and Dentofacial Orthopedics 1990; 97: 463-471.
20.Macfarlane TV, Kenealy P, Kingdon HA, et al. Twenty-year cohort
study of health gain from orthodontic treatment: temporomandibular disorders. Am J Orthod Dentofacial Orthop 2009 Jun; 135(6):692.e1-698; discussion 692-693.
21.Sandowsky C. The risk of
orthodontic treatment for producing temromandibular disorders. Journal of
Dental Education 1992; 49: 398-405.
22.Thialander B, Rubio G, Pena L, De Mayorga C. Prevalence of
tempromandibular dysfunction and its association with malocclusion in children
and adolescents: an epidemic study related to specific stages of dental
development. Angle Orthodontist 2002; 72 (2): 146- 154.
23.Kopp S. Constancy of clinical signs in patients with
mandibular dysfunction. Community Dentistry and Oral Epidemiology 1979;
5: 94-98.
24.
Houston W. the analysis of errors
in orthodontic measurements. American Journal of Orthodontics 1982; 5: 282-
290.
25.Jordan Department of Statistics. Statistical Year
Book. Amman, Jordan:
Jordan Department of Statistics; 2004.
26.Nilner M, Lasting D. Prevalence of functional
disturbances and diseases of the stomatognathic system in 7-14 year olds. Swedish
Dental Journal 1981; 15: 173-183.
27.Glass EG, McGlynn FD, Glaros AG, et al. Prevalence of tembromandibular disorder
symptoms in a major metropolitan area. Craniology 1993; 11: 217- 220.
28.De-Kanter RJ, Truin GJ, Burgerdigik RC, et al. Prevalence in Dutch
adult population and meta-analysis of signs and symptoms of tempromandibular
disorder. Journal of Dental Research 1993; 72: 1509-1518.
29.Gonçalves DA, Dal Fabbro AL, Campos JA, et al. Symptoms
of temporomandibular disorders in the population: an epidemiological study. Journal
of Orofacial Pain 2010; 24 (3): 270- 278.
30.Tecco S, Crincoli V, Di Bisceglie B, et al. Signs and symptoms of
temporomandibular joint disorders in Caucasian children and adolescents. Cranio 2011 Jan; 29 (1): 71- 79.
31.Probert TC, Wiesenfeld D, Read PC. Tempromandibular pain dysfunction
disorder
resulting
from road traffic accidents- An Australian study. International Journal of
Oral Maxillofacial Surgery 1994; 23: 338-341.
32.Levitt SR, McKinney
MW.
Validation of the TMJ scale in the national sample of 10,000patients:
demographic and epidemiological characteristics. Journal of Orofacial Pain
1994; 8: 25-35.
33.Abedel-Hakem AM. Stomatognathic
dysfunction in the western desert
of Egyptian epidemiologic
survey. J Oral Rehab 1983; 10: 461- 468.
34.Nilner M. Functional
disturbances and diseases of stomatognathic system: A cross sectional study. J
Pedodon 1986; 10:
211- 238.
35.Mazengo MC, Kirveskari P. Prevalence of cranio-mandibular disorders in
adults of Ilala District, Dar-es-Salam,
Tanzania. J
Oral Rehab 1991; 18: 568- 572.
36.Carlsson GE. Epidemiological studies
of signs and symptoms of tempromandibular joint-pain-dysfunction. A literature review.
Australian Prosthetic Society Bulliten 1984; 14: 7- 12.
37.Greene CS, Lerman MD, Suture HD, Laskin DM. The TMJ dysfunction
syndrome: heterogenecity of the pain populatin. Journal of American Dental
Association 1969; 79: 1168-1172.
38. Isong
U, Gansky SA, Plesh O. Tempromandibular joint and muscle disorders-type pain
in U.S.
adults: National health interview survey. J Orofac Pain 2008; 22(4):
3-7-22.
39.Van Der Weele LTH, Dibbet JMH. Hilkimo's index: a scale or just a set of
symptoms. J Oral Rehab 1987; 14: 229- 237.