ABSTRACT
Objective: The main objective of our study was awareness of screening for iron deficiency anemia (IDA) among women admitted to the labor ward at King Hussein Medical Centre (KHMC), Amman, Jordan. Their compliance with prescribed iron supplementation and any side effects of treatment were also assessed.
Methods: The data used in this study was collected from a cross-sectional survey primarily targeting patients admitted to the labour ward at KHMC, Amman during the period between February-August 2016. A total of 400 Women were interviewed. A questionnaire was filled after taking the consent of the woman
Results: Anemia was as high as 40% at booking. Sixty-three percent of patients were not compliant with treatment. The most common two reasons for non-compliance were nausea (17%) and constipation (13%). More than half of those treated were still anemic at term
Conclusion: Anemia is a common problem among our patients. We should have guidelines and protocols in place for the management of anemia in pregnancy. More time should be allocated For explaining the risks of anemia, medications and the side effects to ensure compliance. Serum Ferritin should be measured routinely in all patients with low MCV to confirm the diagnosis of IDA.
Key Words: Anemia, Compliance, Perception, Screening, Side-effect of treatment.
JRMS Aug 2018; 25(2):12-18/DOI: 10.12816/0049829
Introduction
The World Health Organization defines anemia in pregnant women as hemoglobin (Hb) concentration of less than 11g/dl in the first trimester, less than 10.5g/dl in the second trimester and less than 10g/dl in the third trimester. (1). Anemia affects 1.62 billion people globally which accounts to 24% of the world population. (2) In the developing world, anemia is a common problem in pregnancy because of the increased demand of iron( iron demand) ,needed to accommodate the increasing maternal blood volume and iron needs for the fetus (2).It is likely to be nutritional.(3) Iron deficiency anemia (IDA) is the leading cause of anemia among pregnant women globally followed by folic acid deficiency despite all the national recommendations and guidelines for treatment and prevention(4). Its main etiologies are a diet poor in absorbable iron, an increased requirement for iron especially during pregnancy, a loss of iron due to parasitic infections, particularly hookworm, and repeated child births.(5) In a typical gestation with a single fetus, the need for an additional 1000 mg of iron is induced by the pregnancy. This amount is often exceeds the iron stores of most women. Diet alone is not enough to supply the body with its needs of iron, (6, 7). Iron deficiency has negative impact on the mother and fetus. It is responsible for various adverse obstetric outcomes due to inability to cope with the stress of child birth. This is ranging from cardiac failure, puerperal sepsis, decreased lactation, preterm baby and low birth weight baby as well as cognitive problems(7).
The prevalence of iron deficiency anemia (IDA) in developing countries is estimated around 52% (8).In Jordan the figure reaches 56.7%, in study done in 2012(9). Our study concentrates on the following points:
• Is there screening for IDA during antenatal care? Are pregnant women being counseled about the benefits and the side effects of the iron supplement?
• Do our pregnant women take their supplement on daily basis?
• Are they compliant with the treatment, and if not, why?
The data collected for this study was collected from a cross-sectional survey primarily targeting patients admitted to the labor ward at King Hussein Medical Center during the period between February-August 2016. A total of 400 Women were interviewed. A questionnaire was filled after the consent of the patient,(Figure 1).It was completed by an Obstetrics and Gynecology resident. The questionnaire sought information about socio demographic characteristics that included age, educational level for women, and area of residence. General obstetric characteristics including gravidity, parity, and birth weight were recorded ,as were past obstetric history of anemia, its previous management and family history of anemia. There were questions regarding haemoglobin levels at booking and at delivery, most common symptoms of anemia, compliance with oral iron supplements and any side –effects of treatment. Patients were also asked about history of receiving intravenous iron or blood transfusion. Birth outcomes were also noted.
Methods
Fig1: Questionnaire sample
Date:
Name: Hospital Number:
Age: Parity: Education :
Area of residence: Family
history of anemia :
Smoking:
History of anemia during previous pregnancies and its treatment:
Gestational age at delivery:
Hb level at interview:
If anemic, most common symptoms:
Hb level at booking :
Compliance:
Side effects:
Any history of IV iron or blood transfusion inthis pregnancy:
Birth outcome:
Results
Most of our patients were in the (25-29) years age group (39%), followed by (20-24) years age group (24%). (Chart1). Most of the women were primigravidas followed by the group of parity 1-2 ,(chart 2). Thirty-five percent of our patients were university graduates and are employed. Anemia was defined as less than 10g/dl was as high as 40% at booking. More than half of those patients continue to have anemia at term (25%). Less than one third of them reported anemia symptoms like dizziness, fatigue and general weakness. Most of the women took oral iron supplements prescribed by doctors at the Royal Medical Services (RMS). Twenty-six percent of them took it more than once daily,(Chart 3). Sixty-three percent of patients were not compliant with treatment. The most common 2 reasons for non-compliance were nausea (17%) and constipation (13%).Thirty-seven percent of those reporting nausea discontinued therapy,(Table I). One hundred and twenty women did not take any iron ,(Chart 3).Forty patients received at least 2 units of blood during their pregnancy or postpartum,15% of anemic mothers had low birth babies,80% of the women interviewed didn’t have counseling about the benefits and side effects of the supplement. All patients had their Hb checked at booking and at delivery, 15% had another check when they presented with symptoms of anemia or bleeding. Almost all of the women received ferrous sulfate as oral iron supplement.
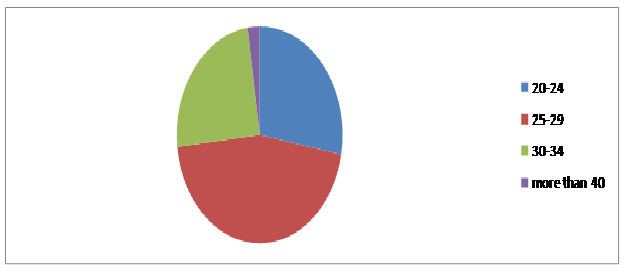
Chart 1: Age distribution in the studied sample.
Chart 2: Distribution of parity in the studied sample
Chart 3: frequency of iron supplementation in the studied sample.
Table I: Side effect.
|
Side
effect
|
Number of patients
|
|
Nausea
and vomiting
|
68=17%
|
|
constipation
|
52=13%
|
|
Epigastric
pain and discomfort
Black
tarry stool
|
40=10%
40=12%
|
Discussion
UP to 52% of pregnant women suffer from anemia in developing countries with great impact on their quality of their lives. If not treated, it can cause maternal and fetal adverse out comes,(10). In our study, screening of hemoglobin level was done at booking visit which is in first trimester, and at delivery. Anemia was diagnosed in our patients if the HB is less than 10 g/dl. Ferritin levels were not requested routinely. MCV was noted in all patients. Recommendations for current practice in the UK are to assess the mother’s hemoglobin concentration at booking and at 28 weeks’ gestation (NICE 2008) and ensure there are systems in place to follow up abnormal results. (11). this is difficult in our country as we do not have clear protocols and the antenatal care is not well established in the primary care system. We should implement guidelines to make sure that anemia among our patients is not missed. Checking Hb in the third trimester is essential because the demand of iron increased to 7.5 mg/day. (12,13,14) this predisposes to postpartum iron deficiency, which is associated with decreased physical abilities , psychic disturbances including emotional instability, depression, stress, and reduced cognitive performance, (15,16,17). In addition, mortality rate was higher in women with very low hemoglobin levels. Also,co-morbidities such as postpartum hemorrhage, acute kidney injury, and disseminated intravascular coagulation were higher in anemic mothers, (18). It is estimated that close to 20% of maternal mortalities are directly linked to anemia and another 50% of maternal deaths are associated with anemia (19).Anemia is common among our patients but no mortalities were noted among the study group. Less than one third of our sample reported symptoms of anemia. The World Health Organization (WHO) and Centers for Disease Control (CDC) technical consultation on the assessment of iron status at the population level concluded that Hb and ferritin were the most efficient combination of indicators for monitoring changes in the iron status of a population as a consequence of iron supplementation (WHO/CDC 2005) but in our study the diagnosis of anemia was based on HB levels and on MCV readings as ferritin levels were not done routinely. Ninety percent of our women in the study were not informed about the details of the iron supplement and its benefits, when and how to take it and side effects. They did not know that the daily diet contains a number of substances (e.g., calcium, polyphenols, and phytates) that inhibit the absorption of iron by approximately 40% (20). Consequently, ferrous iron supplements should be taken between meals, preferably with fruit juice containing vitamin C, which enhances absorption, where as milk, coffee, and tea inhibit absorption (18)(21).All women should be given dietary information to maximize iron intake and absorption, and physicians should give more time to councel patients regarding the benefits of iron supplements. Counseling was poor among the population of ladies we looked at, and we as health care providers should allocate more time and the skills to counsel patients about anemia, treatment and the side effects. From our study, 63% were non-compliant to oral iron supplement and the most common reasons for non-compliance were: nausea (17%) and constipation (13%). So gastrointestinal manifestations are the main factors for non-compliance which led to 37% of our ladies discontinuing treatment. These side-effects may be dose dependent. It is generally considered that doses ≤ 50–60 mg iron/day generate less side-effects than higher doses and that iron given in controlled release formulations is better tolerated (4)(22)(23). However, the meta-regression analysis shows that there is no statistically significant dose-response effect. A study of Danish pregnant women evaluated the effect of 20, 40, 60, and 80 mg ferrous iron daily from 18 weeks gestation to delivery. It appeared that a dose of 20 mg ferrous iron was inadequate to prevent iron deficiency in a substantial number of women. However, 40 mg ferrous iron prevented IDA in more than 95% of the women. Furthermore, there were no significant differences in iron status between women taking 40, 60, and 80 mg elemental iron (8).The Iron preparations that are available in our hospital contains 60 mg of elemental iron. Seventy percent of our pregnant women were taking the supplements daily, 13% were taking it more than once . In the United Kingdom, iron supplements are not given routinely. When the level of ferritin is below 30 microgram/l this should prompt treatment. If ferritin is 70microgram /l this reflects iron stores larger than 500 mg,so there is no need for treatment,(24).In our patients, supplements were given to all patients as ferritin levels were not checked routinely and anemia was common( 40% of women at booking).Twenty five percent (25%) of our patients were anemic at the time of delivery but if they have been treated with iron supplements and they were compliant, then this percentage most likely would have been lower. As found after analyzing the questionnaire, 15% of anemic mothers have low birth babies defined as less than 1.5KG. A meta-analysis summarizing 48 randomized controlled trials and 44 cohort found that, for each 1 g/L increase in maternal hemoglobin, neonatal birth weight increased by 14.0g (6.8 to 21.8g) [10]so that should let us focus on detecting and preventing anemia especially in the third trimester of pregnancy. The Gravidity is an important variable significantly associated with anemia. The risk of anemia increases as the number of pregnancies increases from 3–5 pregnancies (24), but it is even common in the women who had less than three pregnancies. This finding is consistent with other studies conducted in Saudi Arabia and India, in which they found that increased number of pregnancies and deliveries is positively associated with the high risk of developing anemia.(25,26) This is true among our patients as most of them are multiparas. In our study, anemia at booking was found in 40% of our patients compared to the developing countries which is 51% [WHO2004]. This is really high and all these women should have been checked pre-conceptually. Guidelines are essential to tackle this problem to make sure that the Hb of these ladies is within normal limits before conception. Anemia may be relative (physiological) or absolute (27).This may be due to the low socioeconomic status of some of our patients, this will directly affect the nutritional status and iron stores which as a result will decrease. This might explain the high prevalence of anemia among our pregnant population. Forty patients received 2 units of blood before delivery with all the risks of blood transfusions, this is mainly because they presented late in pregnancy with low HB. No patient received IV iron in our study group. This is something we need to look at and encourage those ladies to take this treatment. Most of our patients had IDA based on Hb and MCV results. We need to implement a screening program for IDA. More time needs to be spent with our patients explaining the treatment and the side effects of this treatment. This will ensure and increase the compliance with the medications.
Conclusion
Anemia is a common problem among our patients. Guidelines and protocols for the management of anemia in pregnancy should be developed, implemented and audited. The guidelines should consider both the types of oral iron supplements and recommended doses. More time should be spent with our patients explaining the risks of anemia, supplement options and the associated side-effects in order to maximize compliance. Measurement of ferritin has to be part of the routine assessment of all patients with anemia.
Limitation of the study
It’s a retrospective study and the sample size is relatively small.
References
1.WHO. Archived:Iron deficiency anaemia:assessment,prevention and control: A guide for programme managers,2001:WHO/NHD/01.3
2.Trivedi SS, Puri M. New Delhi: Jaypee Brothers Medical Publishers (P) Ltd; 2010. Anaemia in Pregnancy: Magnitude of Problem, Pharmaceutical; pp. 5–6. Reprint ed.
3.Geneva: WHO; 1989. [Last accessed 2013 Apr 14]. WHO. Preventing and Controlling Anaemia through Primary Health Care: a Guide for Health Administrators and Programme Managers.Availablefrom:http://www.who.int/nutrition/publications/micronutrients/anaemia_iron_deficiency/9241542497.pdf .
4.McDiarmid T, Johnson ED (2002) Clinical inquiries. Are any oral iron formulations better tolerated than ferrous sulfate? J FamPract .2002June;51(6):575-577.
5.Crompton DW, Nesheim MC. Nutritional impact of intestinal helminthiasis during the human life cycle. Annual Review of Nutrition.2002;22:35-59. [PubMed]
6.Leveno KJ, Cunningham FG, Gant NF, Alexander JM, Bloom SL, Casey BM et al. Williams Manual of Obstetrics. Dallas: McGraw Hill, 2003.
7.Brabin BJ, Hakimi M, Pellertier D. An analysis of anemia and pregnancy-related maternal mortality.JNutr. 2001;131:604S–614S. [PubMed]
8.Milman N, Bergholt T, Eriksen L, et al. Iron prophylaxis during pregnancy - How much iron is needed? A randomized dose-response study of 20–80 mg ferrous iron daily in pregnant women. ActaObstetGynecol Scand.2005 Mar;84(3):238-47.
9.Mohammad A. Salahat ,Abdallah I. Ibrahim. Prevalence of Anemia among Jordanian Pregnant Women and the Effect of Early Pregnancy on Alkaline Phosphatase Activity. Jordan Journal of Biological Sciences.2012;5(1): 65-70.
10.Haider BA, Olofin I, Wang M, Spiegelman D, Ezzati M, Fawzi WW, et al. Anaemia, maternal iron use, and risk of adverse pregnancy outcomes: systematic review and meta-analysis. BMJ. 2013;346:f3443. [PMC free article] [PubMed].
11.National Institute for Health and Care Excellence (NICE). Clinical Guideline 62, Antenatal Care: routine care for the healthy pregnant woman. National Collaborating Centre for Women's and Children's Health (UK). London: RCOG Press. 2008. ISBN-13: 978-1-904752-46-2.
12.Milman N. Iron and pregnancy—a delicate balance. Annals of Hematology. 2006;85(9):559–565. [PubMed]
13.World Health Organization. "Iron deficiency Anaemia: Assessment, prevention and control", WHO/NHD/01.3, 2001.
14.Bothwell TH. Iron requirements in pregnancy and strategies to meet them. American Journal of Clinical Nutrition. 2000;72:257S–264S
15.Milman N. Postpartum anemia I: definition, prevalence, causes, and consequences. Annals of Hematology. 2011;90:1247–1253. [PubMed].
16.Beard JL, Hendricks MK, Perez EM, et al. Maternal iron deficiency anemia affects postpartum emotions and cognition. Journal of Nutrition. 2005;135(2):267–272. [PubMed]
17.Corwin EJ, Murray-Kolb LE, Beard JL. Low hemoglobin level is a risk factor for postpartum depression. Journal of Nutrition. 2003;133(12):4139–4142.
18.Ghimire RH, Ghimire S. Maternal and fetal outcomefollowing severe anaemia in pregnancy: Results from NobelMedical College Teaching Hospital, Biratnagar, Nepal.J Nobel Med Coll. 2013;2(3):22-26. doi: 10.3126/jonmc.v2i1.7668
19.Anand T, Rahi M, Sharma P, Ingle GK. Issues in prevention of iron deficiency anemia in India. Nutrition 2014; 30:764–770.
20.Hallberg L, Rossander L, Skaanberg AB. Phytates and the inhibitory effect of bran on iron absorption in man. American Journal of Clinical Nutrition. 1987;45(5):988–996. [PubMed]
21.Hallberg L, Brune M, Rossander L. Iron absorption in man: ascorbic acid and dose-dependent inhibition by phytate. American Journal of Clinical Nutrition.1989 Feb;49(1):140-144.
22.Souza AI, Batista Filho M, Bresani CC, Ferreira LOC, Figueiroa JN (2009) Adherence and side effects of three ferrous sulfate treatment regimens on anemic pregnant women in clinical trials.CadSauda Publica.2009June;25(6):1225-33.
23.Cad SaudePublica 25: 1225–1233. 54. Liguori L (1993) Iron protein succinylate in the treatment of iron deficiency: controlled, double-blind, multicenter clinical trial on over 1,000 patients. Int J ClinPharmacolTherToxicol 31: 103–123. [PubMed]
24.PavordS,MyersB,RobinsonS,AllardS,Strong J. UK guidelines on the management of iron deficiency in pregnancy. Br J Haematol.2012 Mar:156(5):588-600.
25.Elzahrani SS. Prevalence of iron deficiency anemia amongpregnant women attending antenatal clinics at Al-HadaHospital. Canadian J Med. 2012;3(1):10–14.
26.Vivek RG, Halappanavar AB, Vivek PR, HalkiSB,Maled VS, Deshpande PS. Prevalence of Anemia andits epidemiological. Determinants in Pregnant Women.2012;5(3):216–223.
27.Bukar M, Audu BM, Sadauki HM, Elnafaty AU, Mairiga AG. Prevalence of iron deficiency and megaloblastic anaemia at booking in a secondary Health facilty in North Eastern Nigeria.Nigerian Journal of Medicine. 2009;50(2):33–37