Objectives: This study aim to determine the prevalence of depression among vitamin D deficiency patients.
Methodology: Descriptive cross sectional design was recruited. Sample of 310 patients selected conveniently from out patient's clinics at Royal Rehabilitation Center. Data were collected through baseline sample characteristics sheet (age, gender, body mass index BMI, physical activity, presence of chronic disease, previous history of trauma, and vitamin D serum level) and Beck Depression Inventory (BDI).
Result: In general, mean score of serum vitamin D was 42.2nmol/L and mean score of depression was 17.7 on Beck Depression Inventory. Finding revealed that 69.4% of patients have depression varied from mild to severe depression. Patients who have deficient level of vitamin D (<25nmol/L) were have a higher mean score of depression than those who had insufficient or inadequate level of vitamin D (mean = 28.45, 18.75, and 9.10 respectively). Moreover, multiple linear regression showed that serum vitamin D was the strongest predictors for depression after adjusting other confounding variables (β= -.752, p< .01).
Conclusion: Findings of our study contribute to the body of literatures that depression is prevalent among patients with low vitamin D. Further researches are needed to investigate in depth the causality relationship between vitamin D and depression.
Keywords: Serum vitamin D, Depression, Prevalence, Deficiency
.
JRMS April 2020; 27(1): 10.12816/0055466
Introduction
Despite all the medical development that occur worldwide, vitamin D deficiency still consider as a global health problem that affect all age groups (1,2). In 2008, Hollick and Chen estimated over one billion people around the world who had vitamin d deficiency or insufficiency (3). Children, older people and pregnant women are a high risk groups for vitamin d deficiency, but in Middle East it's also highly prevalent among adult group (4). Hospital based study conducted at AL Bashir hospital in Amman in 2010 and 2011 on 3,731 newborns revealed that 94.1% of newborns were vitamin D deficient (5). Another study in Jordan conclude that 22.2% of total Jordanian population above age of 18 had vitamin D deficiency [25(OH) D < 30 ng/ml] (6).
Vitamin D (sunshine vitamin) is 25-hydroxyvitamin D (25(OH) D), fat soluble hormone, and consider as a major circulating form of vitamin D needed for calcium homeostasis and mineralization of bones (7). Human body obtained vitamin D (cholecalciferol) through cutaneous synthesis (result from sun exposure) and dietary intake (8).
Many causes are associated with vitamin D deficiency; people with dark skin have a higher level of melanin concentration than lightened skin. Increase melanin concentration scatters more ultraviolet radiation (UVR-B) resulting in a less efficient conversion of 7-dehydrocholesterol to previtamin D3 (9). Chronic kidney disease (CKD) also affected level of vitamin D. Thus patients have decrease quantities of 1-α-hydroxylase that are available to convert vitamin D to its active form (25-dihydroxyvitamin D) (10). Many medical conditions had been linked with vitamin D deficiency like cystic fibrosis, and celiac disease, crohn's disease. Theses medical conditions are associated with fat malabsorption, and vitamin D absorption require availability of the fat in the gut because it is a fat soluble vitamin (11). Furthermore, obese people are at risk for vitamin D deficiency (12, 13, 14). This explained by subcutaneous fat which extracted vitamin D from the blood and altering its release into the circulation (15). Anticonvulsants drugs which use as a mood stabilizer for bipolar affective disorder and to treat epileptic seizures have been associated with vitamin D deficiency or insufficiency in long term therapy (16, 17, and 18). Enzyme inducer anticonvulsant drugs such as carbamazepine, phenobarbital and phenytoin increase the activity of liver enzymes, which causes rapidly broken down of vitamin D into inactive forms (19, 20).
Literatures reported that low level of vitamin D associated with many diseases that affect human body; Vitamin D is essential prohormone that is needed for normal absorption of calcium and phosphorus from the gut. So, low level of vitamin D leads to calcium and phosphorus deficiency which is the most common cause of rickets in children and osteomalacia in adult (21,22). Moreover, low level of calcium leads to secondary hyperparathyroidism which affect bone density and increase rate of fracture (23, 24), Cancers, such as prostate cancer, ovarian cancer, and multiple myeloma (25), asthma (26), multiple sclerosis (27), chronic inflammatory illnesses, including autoimmune diseases (28), obstructive lung disease (29), and cardiovascular disease (30).
Furthermore, vitamin D is crucial not only for physical health but for psychological and mental health. A lot of studies revealed that vitamin D deficiency is associated with mental illnesses; schizophrenia, schizoaffective disorder, and depression (31,32, 33). Anglin RE, Samaan Z, Walter SD, & McDonald SD (2013) reviewed 13 observational and randomized controlled trials studies about the link between depression and vitamin D deficiency among 31,000 participants and found that there is a relationship between depression and vitamin d deficiency , but the causal relationship is not clear (34). For instance, a study of 615 young adults in the New Zealand found that there was a modestly higher depression score according to the Centre for Epidemiological Studies Depression Scale (CES-D) for those with lower vitamin D levels, serum 25-hydroxyvitamin D was negatively associated with depression symptoms, and conclude that vitamin D is a predictor of depression symptoms (35). Also, a relationship was found between severity of depression and low serum 25-hydroxyvitamin D levels, which remained significant after adjusting participants' socio-demographics, sunlight, urbanization, lifestyle and health. Of interest, an inversely association between serum 25-hydroxyvitamin level and symptoms severity has been shown (p=.003) (36).
Depression and vitamin D deficiency relationship has also been examined in the depressive population treated with vitamin D supplement (37, 38). In randomized double blind controlled trial among 441 obese subjects with body mass index range between 28-47 kg/m2 who received 20,000 or 40,000 IU vitamin D (cholecalciferol) per week versus placebo for one year. Participants with serum 25(OH) D levels less than 40 nmol/L scored significantly higher depressive traits than those with serum 25(OH)D levels greater or equal 40 nmol/L. Beck Depression Inventory (BDI) score showed significant improvement in the two groups who are given vitamin D supplement, but not in the placebo group after one year of treatment and there was a significant decrease in serum parathyroid hormone in the two vitamin D groups. This indicates possible causal relationship between depression and vitamin D (38). In contrast, depressive symptoms in participants with low and high serum 25-hydroxyvitamin D levels were examined in randomized clinical trial to investigate if supplementation with vitamin D would improved depressive symptoms or not. Result revealed that participants with low serum 25-hydroxyvitamin D were more depressed than others with high serum 25-hydroxyvitamin D according Beck Depression Inventory (BDI), but no significant effect of high-dose vitamin D (40 000 IU vitamin D(3) per week for 6 months) found on depressive symptom scores when compared with placebo (39).
Furthermore, high serum 25-hydroxyvitamin D decreases risk of depression. Jääskeläinen. T et al were Studied the relationship of serum 25-hydroxyvitamin D (25(OH) D) with the prevalence of depressive and anxiety disorders among 5371 individuals in Finland and reported that risk of depression was 35% lower among individuals with vitamin D levels above 56 nmol/L compared to those with vitamin D levels below 34 nmol/L (40).
The relationship between depression and vitamin D deficiency among people with other medical comorbidities has been studied also (41. 42). D. J. Armstrong (2007) investigated the relationship between depression and vitamin D deficiency among 75 Caucasian patients. 13.3% of the patients had vitamin D deficiency, while 56.0% had insufficient levels and 30.7% had normal levels. According to Hospital and Anxiety Depression Scale (HADS), depression was higher for those individuals with vitamin D deficiency (Median = 31) than for those with insufficient (HADS = 22.5) or normal (HADS = 23.5) levels of vitamin D (41). Similar result achieved by Jordi, R et al (2006) in their study of a group of subjects with secondary hyperparathyroidism (n = 21), in whom lower serum vitamin D levels were significantly related to higher scores according to Beck Depression Inventory (BDI) as compared to a control group (42).
Despite positive association between vitamin D deficiency and depression, but the causality and nature of the relationship remain unclear. Strong correlation between vitamin D deficiency and depression doesn't explain whether vitamin D was cause or effect of depression (34, 36). 1,25-dihydroxyvitamin D3 receptor (VDR) is widely distributed in human brain in both neurons and glial cells suggested that vitamin D may have autocrine (within cells signaling) and paracrine (cell to cell communication) properties in the human brain (43).Furthermore, VDR present in the area of the brain (cingulate cortex, thalamus, cerebellum, substantia nigra, amygdala, and hippocampus) which are responsible for express 1α-hydroxylase enzymes able to metabolize 25(OH)D to 1,25(OH)2D3 (43,44). Moreover, cingulate cortex, thalamus, cerebellum, substantia nigra, amygdala, and hippocampus are linked to the development of depression (45, 46). Vitamin D affects immune system through inverse association with inflammatory cytokines IL-6 and IL-1β and increased inflammation might be of importance in the pathophysiology of depression and suicide (47). Stumpf WE, Mueller RA, and Hollis BW (1991) were studied the effect of vitamin D on neurotransmitters and found that vitamin D affect gene expression of monoamine neurotransmitters such as serotonin that has a essential role in development of depression (48).
Finally, literatures showed that there is an association between vitamin D and depression through positive effect of vitamin D therapy on depressive symptoms, low level of vitamin D among depressive patients, distribution of vitamin D receptors (VDR) in the area of the brain that is responsible for development of depression, and effect of vitamin D on regulation of monoamine neurotransmitters such as serotonin. However, the exact causal relationship doesn't clear yet. It has also not been recognized whether vitamin D deficiency cause depression, or whether depression causes vitamin D deficiency.
Methodology
Study Design and Sample Selection:
This study was descriptive cross
sectional study.
Sample frame was developed based on non probability sampling method that was
convenient selection according to the inclusion and exclusion criteria of the
study.
Sample was recruited from
out patient's clinics at Royal Rehabilitation Center. Inclusion criteria was all patient with low level of
vitamin D for both gender, while the researchers excluded patients less than 16
years old, postnatal women, patients who had previous history of mental
illness, and patients with body mass index above 30. Data collection was
carried out between June and July, 2018. 310 patients met the criteria of the
study and agreed to participate in the study.
Baseline
characteristics of the study participants:
Demographical
characteristics that were expected by the researchers to be confounding factors
included age, gender, marital status, body mass index (BMI), physical
activity, presence of chronic disease, previous history of trauma. Age was divided into five
groups: 16-26, 27-36, 37-46, 47-56, and >57 years. Marital status was
divided into two groups (single or married). Physical activity was categorized as:
> or equal 3 times per week, 1-2 times per week, < once per week.
Presence of chronic disease was divided into two groups (yes or no). Previous
history of trauma (physical or psychological trauma last 3 month) was divided
into two groups (yes or no).
Body
Mass Index Categorization:
Body
mass index (BMI) was calculated as weight (kg) divided by height squared (m2).
BMI was categorized according to Centers for Disease Control and Prevention
(CDC) as: below 18.5 is underweight, 18.5-24.9 is normal, 25.0- 29.9 is
overweight and 30.0 and above is obese (49).
Vitamin D Categorization:
Vitamin
D concentration was categorized according to International Osteoporosis
Foundation that define different categories for vitamin D (25-hydroxyvitamin D)
status as: Vitamin D deficient < 25
nmol/L, insufficient 25-49 nmol/L, inadequate 50-74 nmol/L, and desirable level
> or equal75 nmol/L (50,51).
Depression
Assessment
A
psychiatrist interviewed the patients to diagnose the presence of depression
based on Diagnostic and
Statistical Manual of Mental Disorders (DSM-5) criteria. Depression
severity among vitamin D deficiency patients were assessed by Beck Depression
Inventory BDI-II (Beck et al. in 1961), followed by BDI-II
is a 21- items on a 4-point scale from 0 (symptom absent) to 3
(severe symptoms). It is self-reporting questionnaire for evaluating the
severity and quantifying levels of depression in normal and psychiatric
populations. Self administration takes around 5–10 min. The recall period for
the BDI is 2 weeks for major depressive symptoms. Scoring is achieved by adding
the highest ratings for all 21 items. The minimum score is 0 and maximum score
is 63. Higher scores indicate greater symptom severity. Scores of 0–10 (considered
normal), 11-16 (Mild mood disturbance), 17-20 (Borderline clinical depression),
21-30 (Moderate depression), 31-40 (Severe depression), and over 40 (Extreme
depression) (52). Beck’s study reported
a coefficient alpha rating of .92 for outpatients and .93 for college student
samples (53).
Ethical Aspect:
This
study was approved by the Ethical Review Board at Royal Medical Services. Each
participant selected in this study provided their written informed consent
which includes study information (title of the study, aim of the study,
risk and benefit, identifying information will keep strictly confidential and
the data will use only
For
scientific purposes, and right to participate or quit the study any time). Data
collection and physical measurement were carried out by rheumatologists,
psychiatrist, and trained nurses.
Data
Analysis:
Participant's
characteristics were described by mean and standard deviation for continuous
variables and frequency and percentage for categorical variables. Chi square X2
tests were performed to determine difference in baseline characteristics of the
study participants (age, gender, marital status, body mass index (BMI), and
previous history of trauma, presence of chronic diseases, physical activity,
and smoking status groups). One Way
ANOVAs was used to compare mean score of depression with regard to vitamin D
categories. Moreover, One Way
ANOVAs was used to compare mean score of depression with regard to gender and
age groups. Multiple linear
regression analysis was conducted to investigate the association between
vitamin D and depression with control the confounding variable. Age, body mass
index (BMI), vitamin D level, and depression score were treated as continuous
or categorical variables according to test used. Analysis was performed using SPSS (version
22) and level of significance was set at P ≤ 0·05.
Result
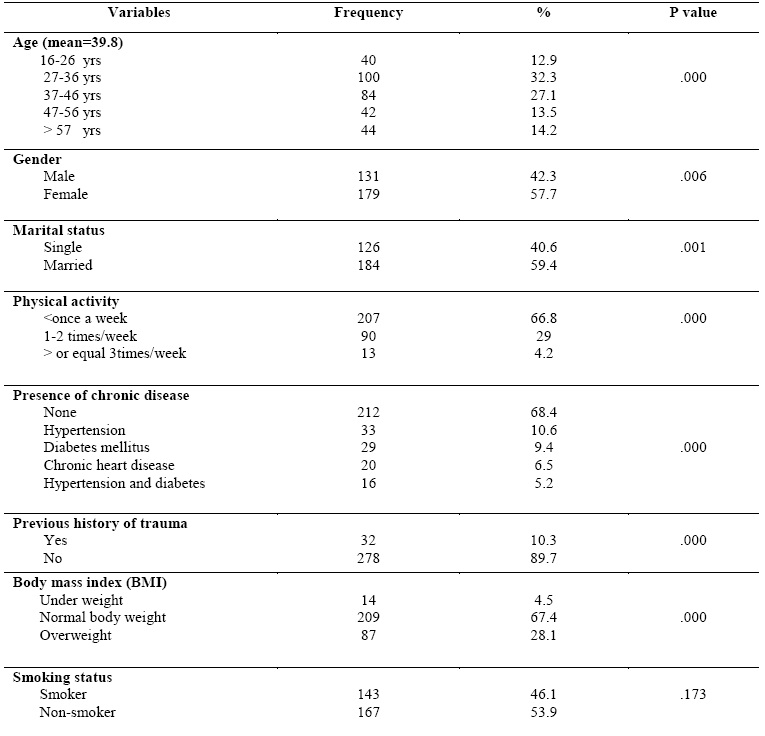
Baseline characteristics of the study
participants: Four hundred and twenty patients were selected for
the study according to low serum 25(OH) D3 (<75nmol/l), and 35 patients were excluded because there body mass index (BMI)
above 30 (obese). Three hundred and ten were consented to participate in the
study (response rate was 80.5%). The mean age of the participants was 38.9
years and the majority of them between ages 27-36 years (32.3%). 179 patients
were female (57.7%). Majority of the participants were practice physical
activity less than once per week (66.8%), 29% practice 1-2 times per week, and
only 4.2% practice three or more times per week. 212 (68.4%) participants
didn’t had a chronic diseases and 32 (10.3%) participants had a previous
history of trauma (physical or psychological trauma). Almost half of the study
participants were smoker (46.1%). With regard to body mass index (BMI),
majority of participants within normal body weight (67.4%) followed by
overweight (28.1%) and underweight (4.5%). Table (I)
Baseline characteristics of the study participants: (Table (1
Baseline
characteristics of the study participants according to vitamin D
level and prevalence
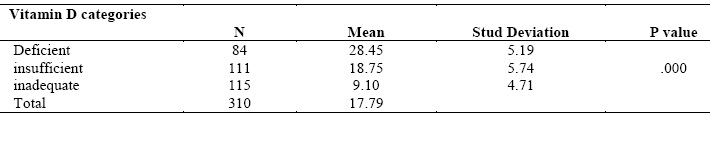
of depression.
Mean score of vitamin D was 42.8 (SD=17.78). 115 participants had
inadequate level of vitamin D (50-74nmol/l), 111 had insufficient level of
vitamin D (25-49nmol/l), while 84 participant had deficient level of vitamin D
(<25nmol/l). Regarding depression among participants, mean score of
depression according to Beck Depression Inventory (BDI) was 17.78 (Borderline
clinical depression). 69.4% of patients have depression varied from mild to
severe depression. 95 participants didn’t have depression, while 69 participants had
moderate depression and 13 participants had sever depression. Table (II).
Table (II): Baseline characteristics of the study participants according to vitamin D level and prevalence of depression.
Comparison of mean score of depression between vitamin D groups
One Way ANOVAs was performed to compare mean score of depression between
vitamin D groups. Mean score of depression was significantly difference between
vitamin D groups (p=.000). Furthermore, the result showed that decrease
level of vitamin D was accompanied by increase the severity of depression
according to Beck Depression Inventory. Mean score of depression among
participants who had deficient level of vitamin D (mean=28.45) was higher than
participants who had insufficient level of vitamin D and mean score of
depression among participants who had insufficient level of vitamin D (mean=18.75)
higher than participants who had inadequate level of vitamin D. Table (III).
3:Comparison
of mean score of depression between vitamin D groups.Table
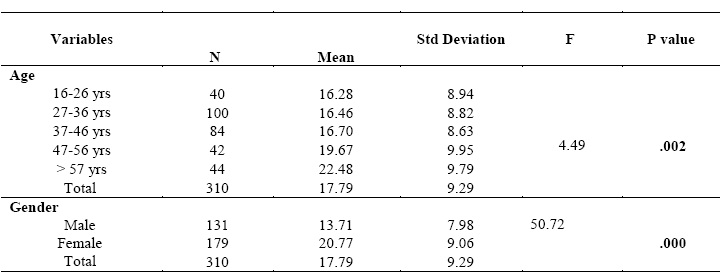
Comparison of mean score of depression with regard to gender and age
groups
Furthermore, One Way ANOVAs was performed to compare mean score of
depression with regard to age and gender. There was a significant difference in
mean score of depression between age groups (p=.002). Age group above 57
years old had the highest mean score of depression, while age group between 16
and 26 years old had the lowest mean score of depression. Moreover, there was a
significant difference in mean score of depression with regard to gender (p=.000).
Mean score of depression among female participants (mean=20.77) was higher than
male (mean=13.71).
Table (IV).
Table (IV):
Comparison of mean score of depression with regard to gender and age groups
Vitamin D deficiency as a predictor for depression
Multiple linear regression analysis was conducted to investigate the association
between vitamin D and depression with control the confounding variable (age,
gender, marital status, physical activity, smoking status, previous history of
trauma, and presence of chronic diseases). Stepwise was used for regressing
previous confounding variables and simultaneously removing those that were not contributed in prediction of depression. Age, marital
status, physical activity, and presence of chronic diseases variables were
excluded. The multiple linear regression model with all five predictors
produced R2 =.768, F (5, 304) =201.2, p <.01.
Vitamin D was significantly negative associated with depression (β= -.862, p<
.01). After adjustment of the other confounding variables (smoking status,
gender, BMI, and previous history of trauma), vitamin D remained significantly
associated with depression (β= -.752, p< .01). As can be seen in (Table V) those with lower
level of vitamin D tend to have higher score of depression.
Table (V):Multivariate
regression analysis for variables predicting depression
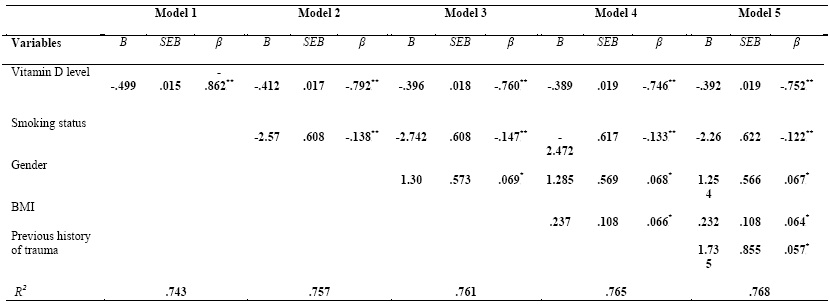
B: unstandardized B, β: standardized β, SEB: standardized error, BMI: Body mass index
Model 1: unadjusted model
Model 2: adjusted for smoking status
Model 3: adjusted for smoking status and gender
Model 4: adjusted for smoking, gender, and BMI
Model 5: adjusted for smoking, gender, BMI, and previous history of trauma
P*< .05, p**< .01
Discussion
Body of literatures about the relationship between vitamin D deficiency and depression are growing, but on the other hand the causality and direction of the relationship still unclear (34). Question about vitamin D deficiency whether it cause depression or effect of depression still not answered. In the last decade, scientific researches concludes that low vitamin D is widespread among people with depression (54, 55), improve depressive symptoms by giving vitamin D supplement (37,38), and increase vitamin D level will decrease the risk of developing depression (40). Ganji. V et al was studied the association between vitamin D and depression among 7970 non-institutionalized US residents and conclude that OR of having current depressive episodes in persons with serum vitamin D ≤ 50 nmol/L is 1.85 fold higher than those with serum vitamin D ≥ 75 nmol/L (P = 0.021) (56).
This study which is the best our knowledge consider first of kind that examined depression among low vitamin D patients in Jordan. Finding of this study revealed that depressive disorder is highly prevalent among patients who had vitamin D deficiency (69.4% of patients have depression varied from mild to severe depression, while only 30.6% didn’t have depression according to Beck Depression Inventory). This result was similar to the result conducted in New Zealand (2014) which revealed that there was a higher depression score according to the Centre for Epidemiological Studies Depression Scale (CES-D) for those with lower vitamin D levels (35).
Furthermore, severity of depression was increased by decrease level of vitamin D. Patients who had deficient level of vitamin D were had a higher mean score of depression than those who had insufficient or inadequate level of vitamin D (mean score of depression equal 28.45, 18.75, and 9.10 respectively). This result was consistent with the result of Milaneschi. Y et al, (2013), Jääskeläinen. T et al (2015), and Von Känel, R (2015) (36, 41, 57).
Gender discrepancy was also found with regard to depression score. Female patients (mean=28.77) had a higher mean score of depression than male patients (mean=18.75). Gender differences with regard to depression well documented in literature. Depression is twice likely prevalent in women than men (58). Moreover, Abate. K reported that the risk of depression is 63% less in male than females (59). Large population based study in Jordan revealed that the prevalence of low vitamin D level less than 30 ng/ml was 37.3% in females compared to 5.1% in males and conclude that dress style that cover most or all of their skin was the main reason (6). This result may support the higher prevalence of depression in female rather than male based on negative association between low vitamin D and depression.
With regard to age, depression score was higher among age group above 57 years old in comparison to other age group less than 57 years. Age related changes in the distribution of depression showed increase in the depressive symptoms score during older age above 60 years old (60). M. Luppa et al analyzed 24 studies that reporting age and gender specific prevalence of depression and estimated the prevalence of major depression in late life to range from 4.6 to 9.3 % and 4.5% to 37.4% for depressive disorders (61).
Significant association had been found between vitamin D levels, age, gender, smoking status, body mass index, previous history of trauma and depression. Vitamin D levels were the strongest predictors for depression. In large cohort study among 1107 participants aged between 18 to 65 years old, association between vitamin D and depression were investigated after controlled of sociodemographic variables. Presence and severity of depressive disorder were associated with low vitamin D suggesting that low vitamin D may represent an underlying biological vulnerability for depression (36).
However, despite of significant relationship between age, presences of chronic diseases, physical activity, marital status variables and depression, but they aren’t contributing in prediction of depression. Age, gender, BMI, and physical activity are factors that previously reported in Jordan to have an effect on vitamin D level (62). Strengths of the current study were the clinical interview (by psychiatrist) in assessing of depressive disorder, that followed by self reported symptoms scale to assess the severity of depressive symptoms that strengthen reliability of the depression diagnosis. Moreover, large sample size may increase the generalizability of the study findings.
Conclusion
Findings of our study
contribute to the body of literatures that depression is prevalent among
patients with low vitamin D. Moreover, severity of depression also affected by
vitamin D level and vitamin D was a significant predictor for depression.
Further researches are needed to investigate the causality relationship between
vitamin D and depression.
Study limitations:
Study limitations included the descriptive cross-sectional design.
Participants were selected by non-random sampling method, so we can't rule out
a selection bias. Causality relationship
between low vitamin D and depression cannot be established because of the
nature of our study. According to Kjaergaard et al. (2012) the relationship between vitamin D and
depression could be bidirectional (39).
Reference
11. Lo CW, Paris PW, Clemens TL, Nolan J, Holick MF. Vitamin D absorption in healthy subjects and in patients with intestinal malabsorption syndromes. Am J Clin Nutr 1985 Oct; 42 (4): 644-649.
12. Vanlint. S. Vitamin D and obesity. Nutrients 2013 Mar 20; 5(3); 949-956.
13. Hyldstrup L, Andersen T, McNair P, Breum L, Transbol I. Bone metabolism in obesity: changes related to severe overweight and dietary weight reduction. Acta Endocrinol 1993 Nov; 129 (5):393-8.
14. WG Tsiaras, MA Weinstock. Factors Influencing Vitamin D Status. Acta Derm Venereol 2011 Mar; 91(2): 115–124.
15. Wortsman J, Matsuoka LY, Chen TC, Lu Z, Holick MF. Decreased bioavailability of vitamin D in obesity. Am J Clin Nutr 2000 Sep; 72 (3): 690-693.
16. G Offermann, V Pinto, R Kruse. Antiepileptic Drugs and Vitamin D Supplementation. Epilepsia 1979 Feb; 1 (20): 3-15.
17. Baek JH, Seo YH, Kim GH, Kim MK, Eun BL. Vitamin D Levels in Children and Adolescents with Antiepileptic Drug Treatment. Yonsei Med J 2014 Mar; 55(2):417-421.
18. Pohan F, Hendarto A, Mangunatmadja I, Gunardi H. Vitamin D levels in epileptic children on long-term anticonvulsant therapy. Paediatr Indones 2015 May; 55(3): 164-70.
19. Farhat G, Yamout B, Mikati AM. Effect of antiepileptic drugs on bone density in ambulatory patients. Neurology 2002 May; 58 (9):1348-1353.
20. Burt R, Freston JW, Tolman KG. The influence of phenobarbital on biotransformation of 25-hydroxycholecalciferol. J Clin Pharmacol 1976 Aug-Sep; 16 (8-9):393-398.
21. Sahay M, Sahay. R. Rickets–vitamin D deficiency and dependency. Indian J Endocrinol Metab 2012; 16(2): 164–176.
22. Holick M. Resurrection of vitamin D deficiency and rickets. J Clin Invest 2006; 116(8): 2062–2072.
23. Henderson JB, Dunnigan MG, McIntosh WB, Abdul-Motaal AA, Gettinby G, Glekin BM. The importance of limited exposure to ultraviolet radiation and dietary factors in the etiology of Asian rickets: a risk-factor model. Q J Med 1987 May; 63 (241): 413–425.
24. Thacher TD, et al. A comparison of calcium, vitamin D, or both for nutritional rickets in Nigerian children. N. Engl. J. Med 1999 Aug; 341(8): 563–568.
25. Edlich R, et al. Scientific documentation of the relationship of vitamin D deficiency and the development of cancer. J Environ Pathol Toxicol Oncol. 2009; 28(2):133-141.
26. Bener A, Ehlayel M, Bener H, Hamid Q. The impact of Vitamin D deficiency on asthma, allergic rhinitis and wheezing in children: An emerging public health problem. J Family Community Med 2014 Dec; 21(3): 154–161.
27. Holick M, Cook M, Suarez G, Rametta M. Vitamin D Deficiency and Possible Role in Multiple Sclerosis. European Neurological Review 2015; 10(2):131–138
28. Agmon-Levin N, Theodor E, Segal RM, Shoenfeld Y. Vitamin D in systemic and organ-specific autoimmune diseases. Clin Rev Allergy Immunol. 2012 Dec 14; 45(2):256-66.
29. Zosky GR, Berry LJ, Elliot JG, James AL, Gorman S, Hart PH. Vitamin D deficiency causes deficits in lung function and alters lung structure. Am J Respir Crit Care Med 2011; 183(10):1336-43.
30. Mozos I, Marginean O. Links between Vitamin D deficiency and cardiovascular diseases. Biomed Res Int 2015 Feb 8; 109275.
31. Sotodeh-Asl N, Tamadon M, Malek F, Zahmatkesh M. Vitamin D deficiency and psychological disorders. J Parathyr Dis 2014 Mar 1; 2(1),21–25.
32. M Bicikova, et al. Vitamin D in Anxiety and Affective Disorders. Physiol. Res 2015 Jun 8; 64 (2): 101-103.
33. Ataie-Jafari A, et al. The association of vitamin D deficiency with psychiatric distress and violence behaviors in Iranian adolescents: the CASPIAN-III study. J Diabetes Metab Disord 2015; 14:62.
34. Anglin RE, Samaan Z, Walter SD, McDonald SD. Vitamin D deficiency and depression in adults: systematic review and meta-analysis. Br J Psychiatry 2013 Feb; 202:100-107.
35. Polak MA, Houghton LA, Reeder AI, Harper MJ, Conner TS. Serum 25-hydroxyvitamin D concentrations and depressive symptoms among young adult men and women. Nutrients. 2014 Oct 28; 6(11):4720-30.
36. Milaneschi Y, et al. The association between low vitamin D and depressive disorders. Mol Psychiatry 2013 Apr 9; 19(4): 444-51.
37. Gloth FM, Alam W, Hollis B. Vitamin D vs broad spectrum phototherapy in the treatment of seasonal affective disorder. J Nutr Health Aging 1999 Jan 1; 3(1):5-7.
38. Jorde R, Sneve M, Figenschau Y, Svartberg J, Waterloo K. Effects of vitamin D supplementation on symptoms of depression in overweight and obese subjects: randomized double blind trial. J Intern Med 2008 Nov 11; 264(6):599-609.
39. Kjaergaard M, et al. Effect of vitamin D supplement on depression scores in people with low levels of serum 25-hydroxyvitamin D: nested case-control study and randomised clinical trial. Br J Psychiatry. 2012 Nov; 201(5):360-8.
40. Jääskeläinen. T et al. Higher serum 25-hydroxyvitamin D concentrations are related to a reduced risk of depression. Br J Nutr 2015 May 14; 14; 113(9):1418-1426.
41. Armstrong DJ, Meenagh GK, Bickle I, et al. Vitamin D deficiency is associated with anxiety and depression in fibromyalgia. Clin. Rheumatol 2007 Apr; 26(4), 551-554.
42. Jorde R, Waterloo K, Saleh F, et al. Neuropsychological function in relation to
serum parathyroid hormone and serum 25-hydroxyvitamin D levels. The Tromso study. J. Neurol, 2006; 253(4): 464-470.
43. Eyles DW, Smith S, Kinobe R, Hewison M, McGrath JJ. Distribution of the vitamin D receptor and 1 alpha-hydroxylase in human brain. J Chem Neuroanat 2005 Jan; 29(1): 21-30.
44. Prufer K, Veenstra TD, Jirikowski GF, et al. Distribution of 1,25-dihydroxyvitamin D3 receptor immunoreactivity in the rat brain and spinal cord. J. Chem. Neuroanatomy 1999; 16(2): 135-145.
45. Price JL, Drevets WC. Neurocircuitry of mood disorders. Neuropsychopharmacology Reviews 2010; 35 (1): 192-216.
46. Sheline YI, Gado MH, Kraemer HC. Untreated depression and hippocampal volume loss. Am J Psych 2003 Aug; 160 (8): 1516-1518.
47. Grudet C, Malm J, Westrin A, Brundin L. Suicidal patients are deficient in vitamin D, associated with a pro-inflammatory status in the blood. Psychoneuroendocrinology 2014 Dec; 50:210-9.
48. Privette TH, Stumpf WE, Mueller RA, Hollis, BW. Serum 1,25-dihydroxyvitamin D3(soltriol) levels influence serotonin levels in the hypothalamus of the rat. Abstr. Soc. Neurosci 1991; 17, 498.
49. Centers for Disease Control and Prevention (2009a). Body Mass Index: Considerations for Practitioners. Available https://www.cdc.gov/obesity/downloads/BMIforPactitioners.pdf (accessed 2 Jun 2018).
50. Hilger J. Friedel A. Herr R, Rausch T, Roos F, Wahl DA, Pierroz DD,Weber P, Hoffmann K. A systematic review of vitamin D status in populations worldwide. Br. J. Nutr 2014 Jan 14; 111 (1); 23–45.
51. Mithal A, Wahl DA, Bonjour JP, Burckhardt P, Dawson-Hughes B, Eisman JA, El-Hajj Fuleihan G, Josse RG, Lips P, Morales-Torres J, et al. Global vitamin D status and determinants of hypovitaminosis D. Osteoporos Int 2009 Nov; 20 (11): 1807–1820.
52. Kendall PC, Hollon SD, Beck AT, Hammen CL, Ingram RE. Issues and recommendations regarding use of the Beck Depression Inventory. Cognitive Ther Res 1987;11 (3): 289–299.
53. Beck AT, Steer RA. Manual for the Beck Depression Inventory. San Antonio, TX: The Psychological Corporation; 1987.
54. Eskandari F, Martinez PE, Torvik S, Phillips TM, Sternberg EM, Mistry S, et al. Low bone mass in premenopausal women with depression. Arch Intern Med 2007 Nov 26; 167 (21): 2329-2336.
55. Nebhinani N, Sharma P, Suthar N. Role of Vitamin D supplementation in patients with depressive disorders and hypovitaminosis D: A longitudinal study. J Mental Health Hum Behav 2017; 22 (1) :14-20.
56. Ganji V, Milone C, Cody MM, McCarty F, Wang YT. Serum Vitamin D concentrations are related to depression in young adult US population: The Third National Health and Nutrition Examination Survey. Int Arch Med 2010 Nov 11; 3 (1):29.
57. von Känel R; Fardad N; Steurer N; Horak N; Hindermann E; Fischer F; Gessler K. Vitamin D Deficiency and Depressive Symptomatology in Psychiatric Patients Hospitalized with a Current Depressive Episode: A Factor Analytic Study. PLOS ONE journal.pone 2015 Sep 23; 10 (9).
58. Patten SB, Wang JL, Williams JV, et al. Descriptive epidemiology of major depression in Canada. Can J Psychiatry 2006;51 (2):84–90.
59. Abate K. Gender disparity in prevalence of depression among patient population: a systemic review. Ethiop J Health Sci 2013 Nov; 23 (3).
60. Tomitaka, et al. Age-related changes in the distributions of depressive symptom items in the general population: a cross-sectional study using the exponential distribution model. PeerJ 2016; 4 (1):e1547.
61. Luppa M, Sikorski C, Luck T, et al.Age- and gender-specific prevalence of depression in latest-life – systematic review and meta-analysis. J Affect Disord 2012 Feb; 136 (3), 212–221.
62. Al-Horani H, et al. Nationality, Gender, Age, and Body Mass Index Influences on Vitamin D Concentration among Elderly Patients and Young Iraqi and Jordanian in Jordan. Biochem Res Int 2016 Mar; (2); 1-8.